The concentrations of sulfur dioxide both emitted into the air and measured at monitoring stations have decreased significantly over the past two decades. Emissions dropped from about 26.4 million short tons (24 million metric tons) annually in 1982 to about 17.6 million short tons (16 million metric tons) in 2001.
The only area in which improvement did not occur was in emissions from transportation, which rose from about 770,000 tons (700,000 metric tons) in 1980 to about 2 million short tons (1.8 million metric tons) in 2000.
Control of sulfur dioxide emissions from stationary sources (such as power plants) usually takes one of three forms: fuel cleaning, also known as fuel beneficiation; removal of sulfur during combustion; or flue gas processing.
The goal of beneficiation is to remove as much sulfur from a fuel as possible before it is ever burned. When burned, fuel with lower sulfur content will produce less sulfur dioxide. Beneficiation is usually accomplished by a physical process that separates one form of sulfur, pyritic sulfur, from coal.
Pyritic sulfur consists of sulfur minerals (primarily sulfides) that are not chemically bonded to coal in any way. The name is taken from the most common form of mineral sulfur usually found in coal, pyrite, or iron sulfide (FeS2).
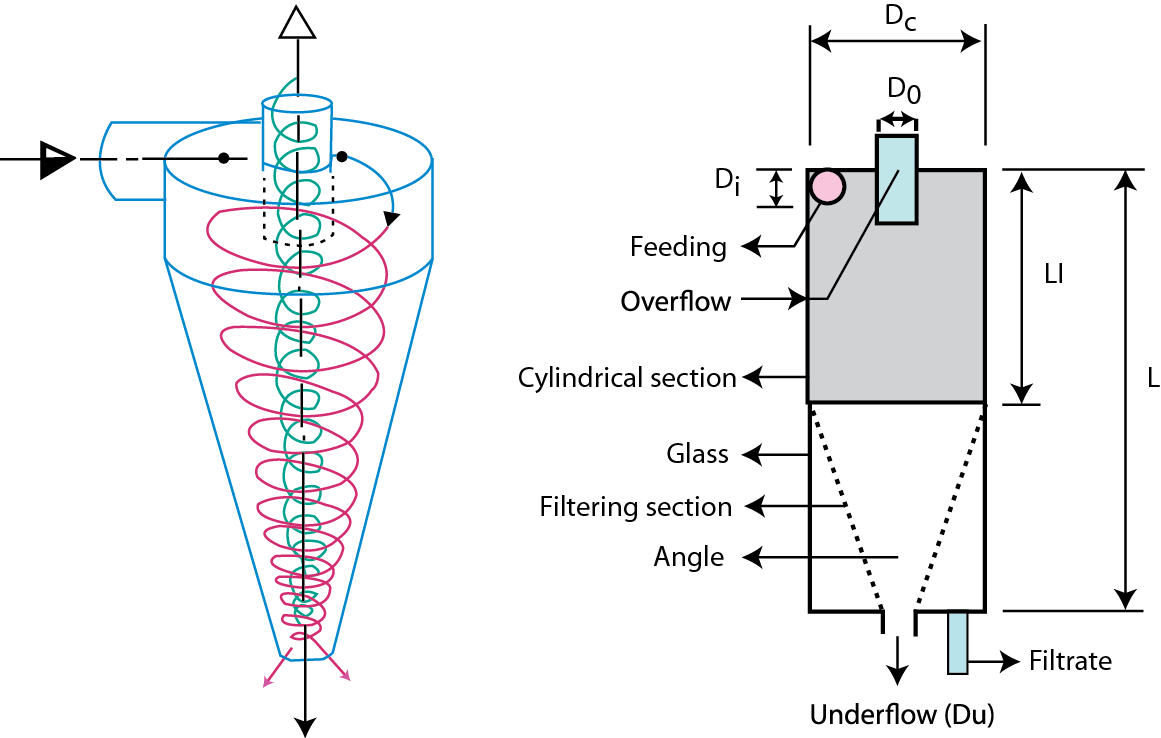
Mineral sulfides have significantly different densities from those of coal, so they can be removed from it by methods that take advantage of this difference, such as hydrocycloning, dry cycloning, and froth floatation.
In hydrocycloning, for example, slurry of coal and water is introduced into a centrifuge-type tank with a central cylinder. As the tank spins, the sulfides are thrown outward against the inner wall of the tank, while the coal is ejected upward and out of the tank through the central cylinder. The operation of a cyclone is similar to that of a hydrocyclone, except that dry coal rather than coal slurry is injected into the device.