The combustion temperature is kept relatively low, in the range of 750°C–925°C, to prevent the formation of oxides of nitrogen. Fluidized bed combustion is generally able to remove up to 95 percent of the sulfur found in coal.
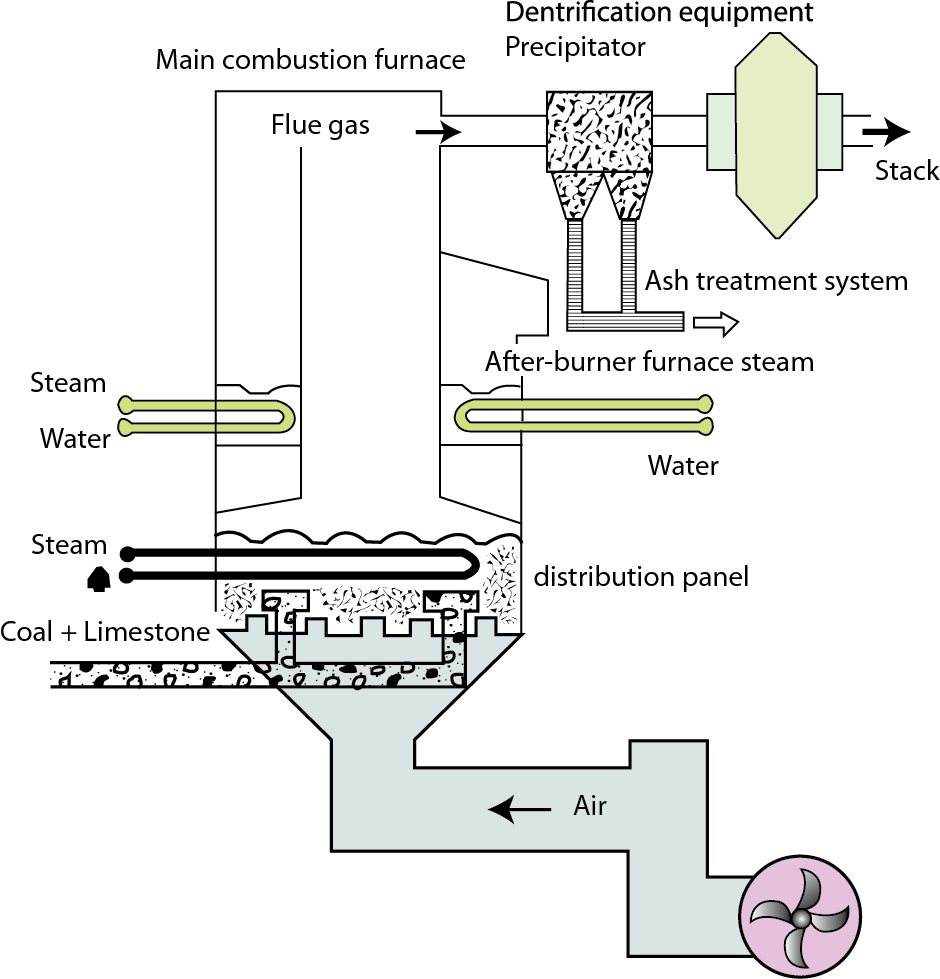
Most post-combustion cleaning systems make use of scrubbers to remove sulfur dioxide.
Scrubbers are devices that contain some chemical that will react with sulfur dioxide in flue gases.
Two kinds of scrubbers are used, wet and dry.
As their names suggest, the two types differ in the extent to which water is mixed with the chemical used to remove sulfur dioxide.
In wet scrubbers, the chemical is dissolved in or mixed with water and then sprayed into flue gases.
In dry scrubbers, the chemical is pulverized and then sprayed into flue gases.
The most common chemical used in scrubbers today is limestone (calcium carbonate; CaCO3) because it reacts with sulfur dioxide readily to form calcium sulfite (CaSO3), which can be collected and sold as gypsum.