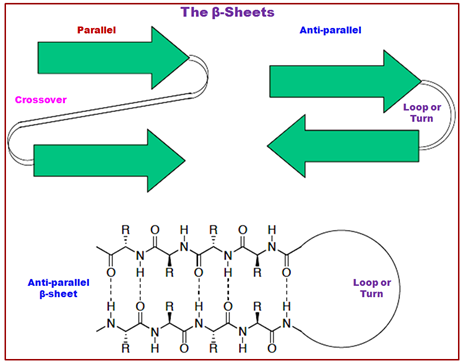
Figure 2.18: The structure of β-sheets-parallel/anti-parallel.
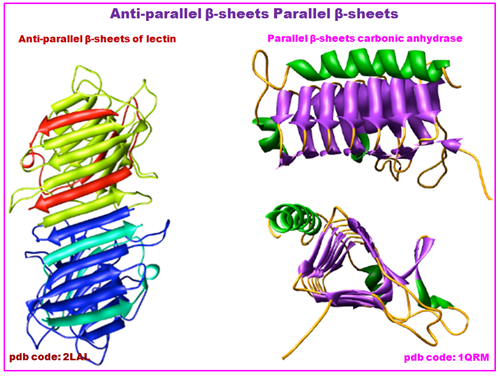
Figure 2.19: The structure of anti-parallel β-sheets of Lectin and parallel b-sheet of Carbonic anhydrase.
2.4.3. β-Turn
A region of the protein involving four consecutive residues where the polypeptide chain folds back on itself by nearly 180°. This chain reversal gives proteins a globular rather than linear structure. Thus a turn may be defined as is a structural motif where the Cα atoms of two residues separated by usually 1 to 5 peptide bonds are in closer in < 7 Å distant while the corresponding residues do not form a regular secondary structure element such as an alpha helix or beta sheet. The backbone dihedral angles are not constant for all the residues in the turn which in contrary to helices.
A turn can be converted into its inverse turn (almost mirror-image turn) by changing the sign on all of its dihedral angles. Thus, the γ-turn has two forms, a classical form with (φ, ψ) dihedral angles of roughly (75°, -65°) and an inverse form with dihedral angles (-75°, 65°). At least eight forms of the β-turn have been identified, varying mainly in whether a cis isomer of a peptide bond is involved and on the dihedral angles of the central two residues. Types VIa1, VIa2 and VIb turns are subject to the additional condition that residue (i + 2)(*) must be a cis-proline. Several turns are listed below in the table 1.
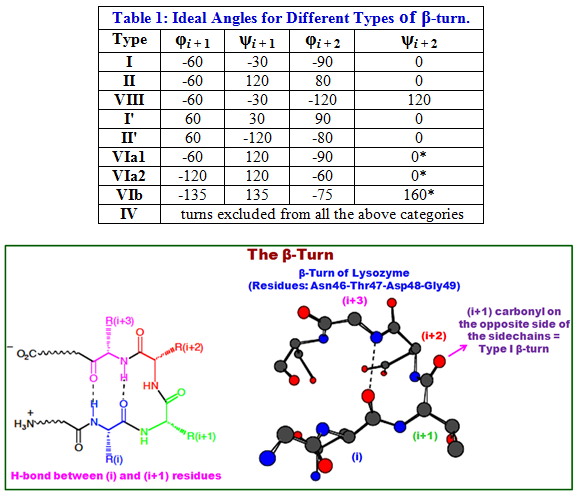
Figure 2.20: The H-bonding in β-turn structure and the β-turn of Lysozyme.