2.4.1. The α-Helix:
- Polypeptides chain folds along with their backbone into regular conformations which would similar to the α-keratin fiber. The most simple and elegant arrangement is a right-handed spiral conformation known as the ‘α-helix’.
- Amino acids wound into a helical structure 3.6 amino acids per coil, 5.4 Å.
- Properties of the α -helix.: The structure repeats itself every 5.4 Å along the helix axis, i.e. the α-helix has a pitch of 5.4 Å. α-helices have 3.6 amino acid residues per turn, i.e. a helix of 36 amino acids long would form 10 turns. The separation of residues along the helix axis is 5.4/3.6 or 1.5 Å, i.e. the α-helix has a rise per residue of 1.5 Å.
- Every main chain C=O and N-H group is hydrogen-bonded to a peptide bond 4 residues away (i.e. Oi to Ni+4). This gives a very regular, stable arrangement.
- The peptide planes are roughly parallel with the helix axis and the dipoles within the helix are aligned, i.e. all C=O groups point in the same direction and all N-H groups point the other way. Side chains point outward from helix axis and are generally oriented towards its N-terminal end.
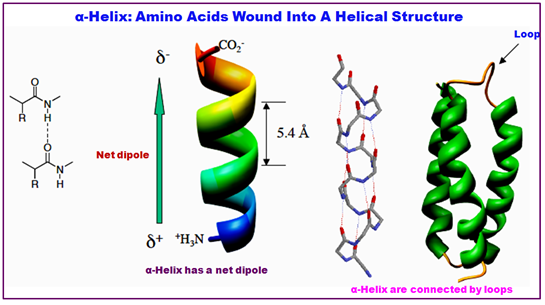
Figure 2.14: The structure of a α-helix.
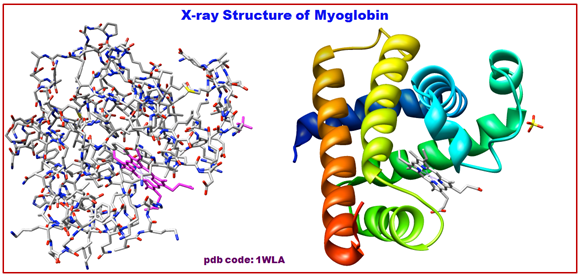
Figure 2.15: The structure of α-helix of Myoglobin.
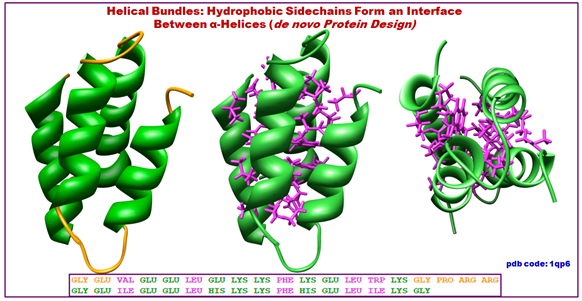
Figure 2.16: The structure of helix bundles.
2.4.2. β-Sheets
The β-sheet or β-pleated sheet is the second form of regular secondary structure in proteins. It is only somewhat less common than alpha helix. Beta sheets consist of beta strands which are connected laterally by at least two or three backbone hydrogen bonds. Thereby it forms a twisted, pleated sheet. A β-strand is a stretch of polypeptide chain with 3- to10-amino acids long with backbone in an almost fully extended conformation. The higher-level association of β-sheets has been implicated in formation of the protein aggregates and fibrils responsible for generating many human diseases, notably the amyloidoses such as Alzheimer's disease.

Figure 2.17: The structure of β-sheets-parallel/anti-parallel.