1.10.3. Chemistry of Living Organisms
So we knew that all living things are composed of one or more cells and the products of those cells. The chemical compounds that make up the structures in cells are a mixture of organic compounds and inorganic compounds. Organic compounds always contain carbon and hydrogen (and maybe some other elements), inorganic compounds do not contain carbon and hydrogen together. Simply, Organic refers to life and inorganic compounds make up non-living substances. Organic compounds are found in living things, their wastes, and their remains. Examples of organic compounds that are basic to life includes: carbohydrates (sugars, starches), lipids (fats & waxes), proteins, enzymes, nucleic acids (DNA & RNA). Examples of inorganic compounds includes: water, carbon dioxide. The elements (atoms) in organic compounds are held together by covalent bonds, which form as a result of the sharing of two electrons between two atoms.
1.10.4. The Composition of Living Cell
All living organism contains the following organic molecules for their lives:
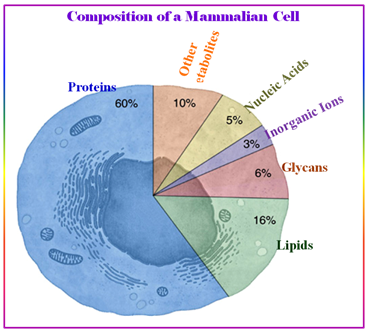
Figure 1.34: The composition of mammalian Cell
1.10.4.1. Carbohydrates as One of the Major Components of Living Organism:
- Main source of energy for living organisms: sugar and starch.
- Sugar is broken down inside the body into glucose, which is used for energy.
- Excess sugar is stored as starch.
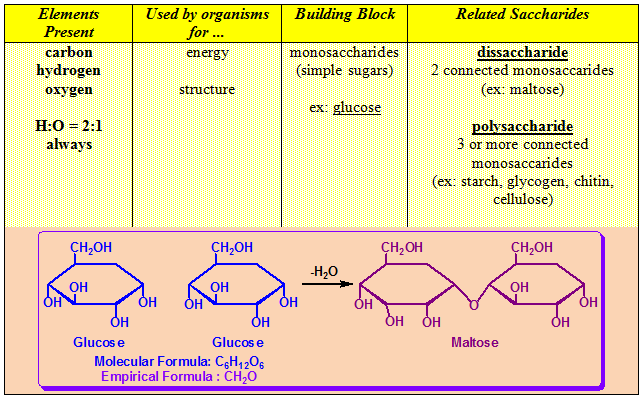
- Chitin and cellulose are examples of carbohydrates with structural functions. Chitin is the material that makes up the exoskeletons of all arthropods (insects, spiders, lobsters, etc.). Cellulose is what the cell wall in plant cells is made of.
- Starch is the form by which plants store extra carbohydrates. Glycogen, sometimes referred to as animal starch, is the form by which animals store extra carbohydrates. We store glycogen in our livers.