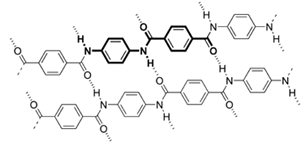
Hydrogen bonds in polymers: structure of Para-aramid:
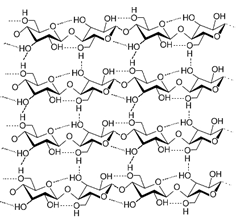
Hydrogen bonds in Sugar: A strand of cellulose (conformation Iα), showing the hydrogen bonds (dashed) within and between cellulose molecules.
H-bond in small organic molecules: An example of intermolecular hydrogen bonding in a self-assembled dimer complex of a Donor-Acceptor molecules.
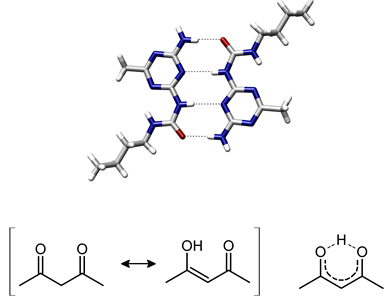
Intramolecular hydrogen bonding in acetylacetone helps stabilize the enol tautomer
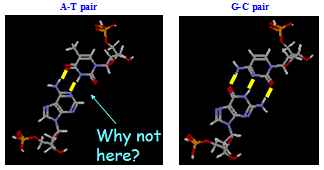
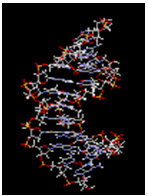
Base Pairs in DNA: H-bonding:
DNA Double helix: H-bonding, π-π stacking interaction, Hydrophobic interactions, Electrostatic interactions:
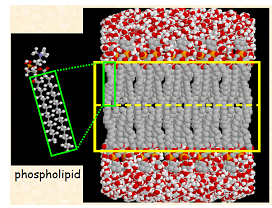
Lipid Bilayer: Induced Dipole – Induced Dipole Interaction for Hydrocarbon Chains
Proteins structure: Hydrophobic interactions, hydrogen bonds and salt bridges though are all relatively weak interactions but the large number in a protein combine to give the overall stability of the structure.
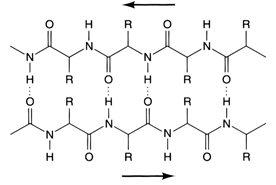
Diagram of β-pleated sheet with H-bonding between protein strands
In addition to these numerous forces, the role of solvation is important and so, consequently, is the relationship between the biocompounds studied and their environments, relationships driven by weak interactions. For example, native globular proteins are mainly constituted of amino acids, some of them buried inside the structure with a low solvent accessibility. In contrast, other amino acids could be at the surface of the protein. The latter are solvated and they play an important role in the intermolecular interactions. Proteins are tightly packed. They have a compact structure with ample intra-globular voids. The voids are small cavities subjected to dynamic fluctuations which can govern conformational dynamics, one source of protein activity. Both voids and weak interactions allow protein fluctuations which can be large, such as rotation of side chains and small protein inducing subcon formations. A protein in solution is in a dynamic and thermodynamic equilibrium of various conformers. The multiple conformations depend also on the nature of the interactions involved. To understand these molecular fluctuations, one way involves perturbing them. This is why it is important to carry out both pressure and temperature experiments on these systems to separate thermal and volume effects.
However, weak interactions are not typical for just bios stems. They are also involved in understanding the pressure effects on many inorganic or bioinorganic reactions: synthesis, solvent exchange, legend substitution, addition, elimination, electron transfer and radiation induced reactions.