Hydrogen bond: The hydrogen bond is a specific and very important type of intermolecular interaction. A hydrogen bond occurs when the electrons forming the covalent bond between the hydrogen atom and the oxygen, nitrogen, or fluorine atom are pulled toward the electronegative atom. Hydrogen bond present in water. As such, it is the strongest of the intermolecular interactive forces.
In summary, weak non-covalent interactions play an important role in many chemical processes. In chemistry, these forces play a crucial role in controlling the shape, selectivity and reactivity. As a matter of fact, the phenomenon of “Molecular Recognition” is dominated by weak forces, which include the H-bonding, electrostatic, stereo-electronic, π−stacking interactions as well as hydrophobic and steric interactions.
These weak non-covalent forces can broadly be grouped into five categories all of which plays an important role in Organic chemistry.
- Electrostatic interactions: Strongest interaction between dipolar and charged molecules.
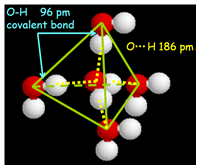
- H-bonds: A hydrogen bond is the attractive interaction of a hydrogen atom with an electronegative atom, like nitrogen, oxygen or fluorine (thus the name "hydrogen bond", which must not be confused with a covalent bond to hydrogen). The hydrogen must be covalently bonded to another electronegative atom to create the bond. These bonds can occur between molecules (intermolecularly), or within different parts of a single molecule (intramolecularly). The hydrogen bond (5 to 30 kJ/mole) is stronger than a van der Waals interaction, but weaker than covalent or ionic bonds. This type of bond occurs in both inorganic molecules such as water and organic molecules.
- Charge transfer complexes: A charge-transfer complex (CT complex) or electron-donor-acceptor complex is a chemical association of two or more molecules, or of different parts of one very large molecule, in which the attraction between the molecules (or parts) is created by an electronic transition into an excited electronic state, such that a fraction of electronic charge is transferred between the molecular entities. The resulting electrostatic attraction provides a stabilizing force for the molecular complex. The source molecule from which the charge is transferred is called the electron donor and the receiving molecule is called the electron acceptor. It is found frequently in flavine chemistry.
- Van der Waals forces: Influenced by polarizability and structure of the molecule. The van der Waals force (or van der Waals interaction) is the attractive or repulsive force between molecules (or between parts of the same molecule) other than those due to covalent bonds or to the electrostatic interaction of ions with one another or with neutral molecules. It includes: (a) force between permanent dipole and a corresponding induced dipole and (b) instantaneous induced dipole-dipole forces (London dispersion force).
- Hydrophobic interactions: The tendency of hydrocarbons (or of lipophilic hydrocarbon-like groups in solutes) to form intermolecular aggregates in an aqueous medium, and analogous intramolecular interactions.
- π-π stacking interaction: Stacking refers to a stacked arrangement of often aromatic molecules, which is adopted due to interatomic interactions. Stacking is often referred to as π-π interaction, though effects due to the presence of a π-orbital are only one source of such interactions, and in many common cases appear not to be the dominant contributors.
Besides the fact that the origin of the intermolecular forces is purely chemical, all of them play a crucial role in biological world. Weak interactions are, in biochemistry, one of the keys to understanding pressure effects on biomaterials. The main reason is because weak interactions are strongly involved in the structure of biomolecules and in the functions of many bioprocesses. Non-covalent forces are actively taking part in (a) chemical and biological molecular recognition world, (b) catalysis in chemical synthesis, prebiotic world, in vivo (how the cell is controlled?) and in (c) chemical models
Native structure of biological compounds, i.e. the conformation that displays biological activity, is the result of a delicate balance between stabilizing and destabilizing interactions (strong or weak) within the polypeptide chains for proteins and with the solvent. Native biocompounds are stable in a narrowphysical–chemical zone depending both on the nature of the biomaterials itself and on the environmental conditions (such as other proteins, nucleic acids, membranes, lipids, solutes, solvents, salts, pH, temperature).