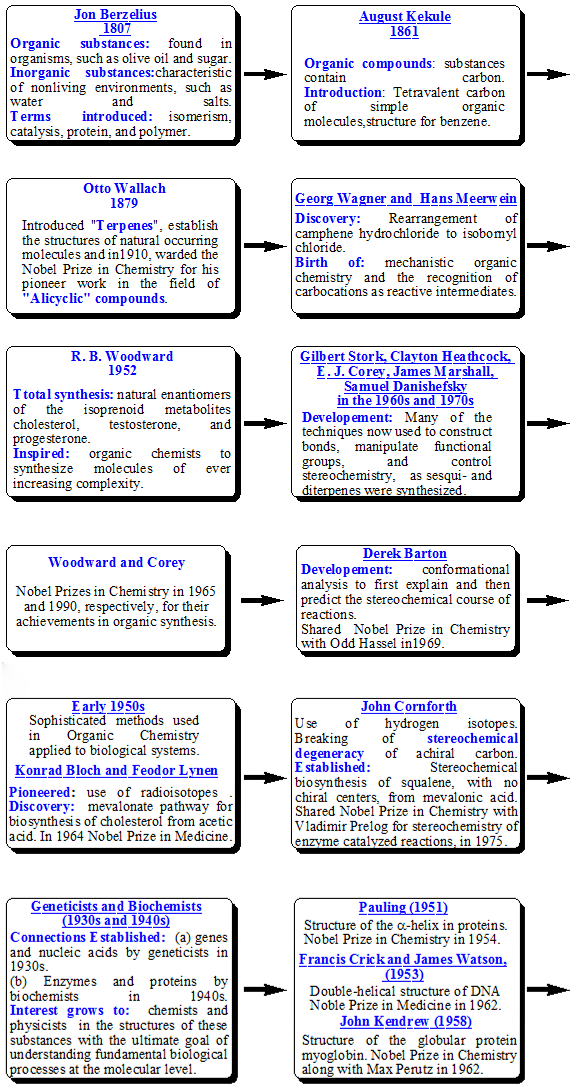
1.6. Historical Connection between Organic and Biological Chemistry:
1.7. Weak Interactions in Organic and Biological World
"Apart from consideration of the hydrogen bond, we organic chemists have really paid little attention to linkages other than the purely covalent. I believe that it will be the duty of organic chemists in the future to study the weak non-bonding interactions which are of enormous importance in the large natural macromolecules. Such studies will lead to a new blossoming of organic chemistry in the future.”
---------Lord Alexander R. Todd
“Chemie in Vergangenheit und Gegenwart” Lecture, on the occasion of the presentation of the “Goldenes Doktordiplom”, Universität Frankfrut October 16, 1981.
1.7.1. Non-covalent Interactions: Chemical origin
The attractions of molecules for each other are called intermolecular interactions to distinguish them from covalent and ionic bonding, forms of intramolecular interactions.
Intermolecular interactions are most significant in liquid and solid phases where molecules are very close together. In fact, even in liquids and solids intermolecular interactions are only strong for molecules that are next to each other. The interactions of molecules in the liquid and solid states have significant consequences that are readily observable. The strength of intermolecular interactions affects numerous properties, including boiling points, miscibility, and solubility.
van der Waals interactions: All molecules interact with other molecules through London dispersion forces, also called van der Waals interactions. London dispersion forces are the attractive forces of one transient dipole for another. As for example in the case of an argon atom in liquid argon, for example, the argon atoms next to the one with temporary dipole would feel the effect of the dipole. Thus the temporary dipoles are propagated through a liquid or solid. The larger the size of atoms and the more electrons they possess, the greater the probability of forming substantial transient dipole interactions. Molecules which are non-polar and non-polar functional groups of molecules only experience London dispersion or van der Waals interactions with other molecules or functional groups.
Polar-polar interaction: Some molecules and functional groups have permanent dipoles that result from a non-symmetric geometric arrangement of atoms of different electronegativity. The portion of a molecule with a permanent dipole will be attracted to the portion of a neighboring molecule with its own permanent dipole. A polar-polar interaction is stronger than a van der Waals interaction.