Chain Transfer Reaction
In this reaction , one radical of the multiplying chain terminates and without the loss of any number of free radicals, a new chain begins. These reactions control the degree of polymerization and the rate of branching of the polymers.
Branching usually occurs at higher temperature, with the reaction of a multimer with a dead chain, followed by lengthening of the branch due to successive monomer addition.
Chain Termination
A chain terminates with the net loss of one free radical. It may occur by the collision of growing chains with coupling or disproportionation. However , if a growing chain collides with an initiation radical, undesirable impurities or the wall of the reaction vessel, then it might also terminate.
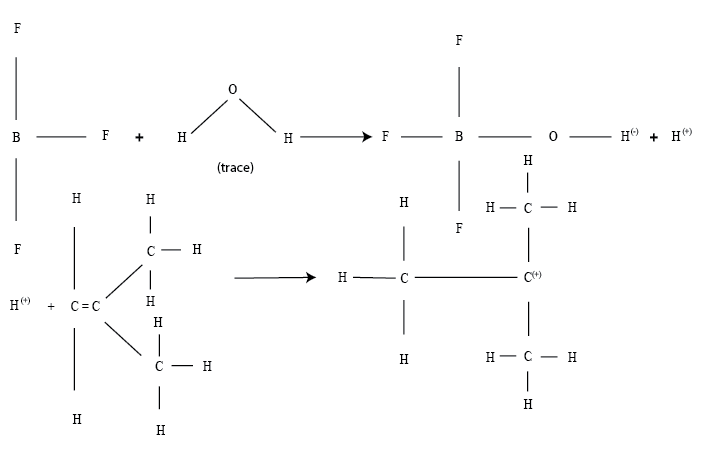
iii .Ionic Reaction :
These are similar to chain addition reaction with free radicals , however ; instead of neutral, unpaired electrons, they have a positive or negative charge. A co-catalyst may be used, which is usually a trace of H2O, which is necessary to produce ionization, as the reaction cannot occur in absolute dry conditions.
An example of ionic addition reaction is the preparation of polyisobutylene with boron trifluoride catalyst.
Initiation