Block Polymerization
In this type, multimers resulting from straight polymerization coupled together in blocks.
![]()
Graft Polymerization
These have a structure which differs from block polymers ; and are formed from M and N multimers.
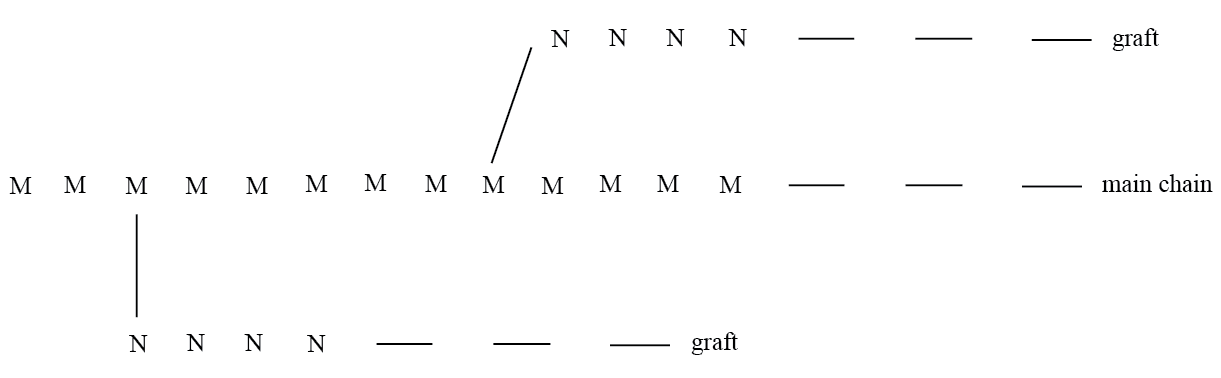
In Graft polymerization, β – or X - radiation or chemically reactive functional groups ensue the formation of reactive radicals on the main chain or on the grafted section.
Reaction Kinetics :
Let,
A – radical source
B – monomer molecule
*- activated
Bm , Bn – growing monomer molecule chain of lengths ‘m' and ‘n'.
X – any solvent , impurity, etc. which can act as transferable energy species.
Initiation
It is the rate – controlling step in the chain Addition Reaction. This stage is relatively slow and requires energy for breaking and forming the bonds. The energy is supplied as thermal energy or in the form of radiations ( α -, β -, or ¥ - rays). However , by using a catalyst, the activation energy required can be substantially lowered.
Propagation
These reactions occur between the already formed free radicals and an inactive monomer. With low activation energy, there is more probability of reaction or collision between the species. As a result, rapid reactions occur to attain the given degree of polymerization.