a. Monofunctional or unifunctional : When a single reactive group is present in the monomer molecule, then it is termed as monofunctional or unifunctional. However a monofunctional group cannot lead to the propagation of a polymer chain. For example , in carboxylic acid, CH3COOH, the –COOH group is the monofunctional group.
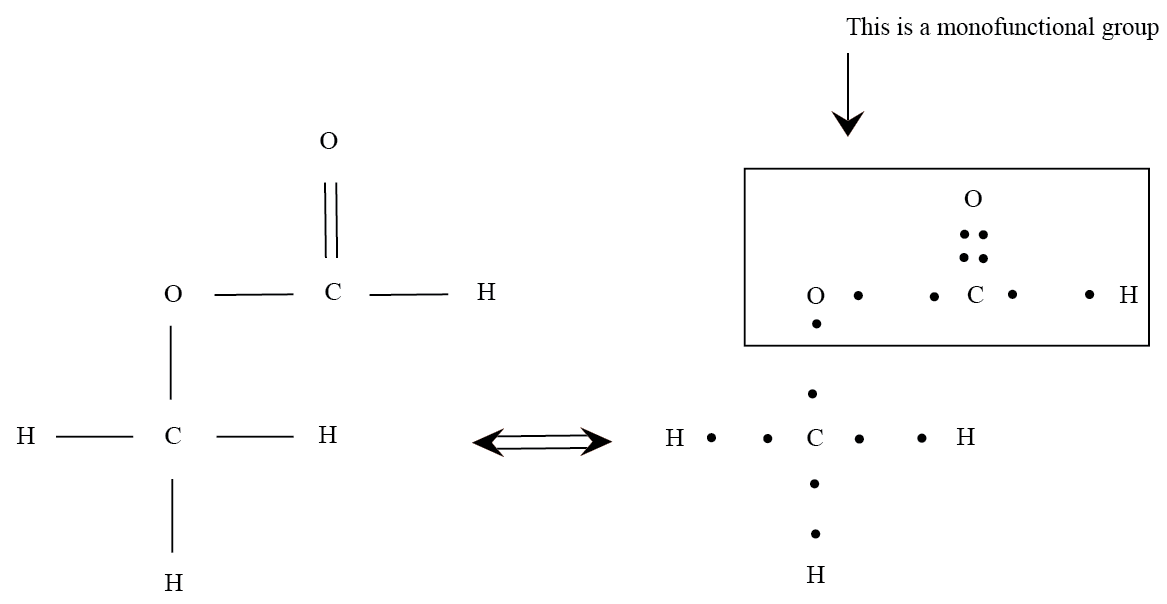
b. Bifunctional : When two reactive groups are present in the monomer molecule, then it is termed as bifunctional. More often than not, polymerization reaction with bifunctinal groups occur when a double bond splits to couple with another double bonded monomer.If a double bonded molecule is present, then the polymer would be –
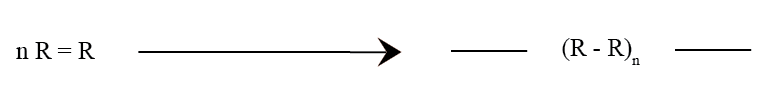
c. Trifunctional : When a monomer contains three groups that are reactive, then it is termed as trifunctional. It is important to note that a trifunctional polymer may be a mixture of monofunctional and bifunctional monomer structures.
Let us consider the reaction of phenol and formaldehyde to form trimethyl phenol.
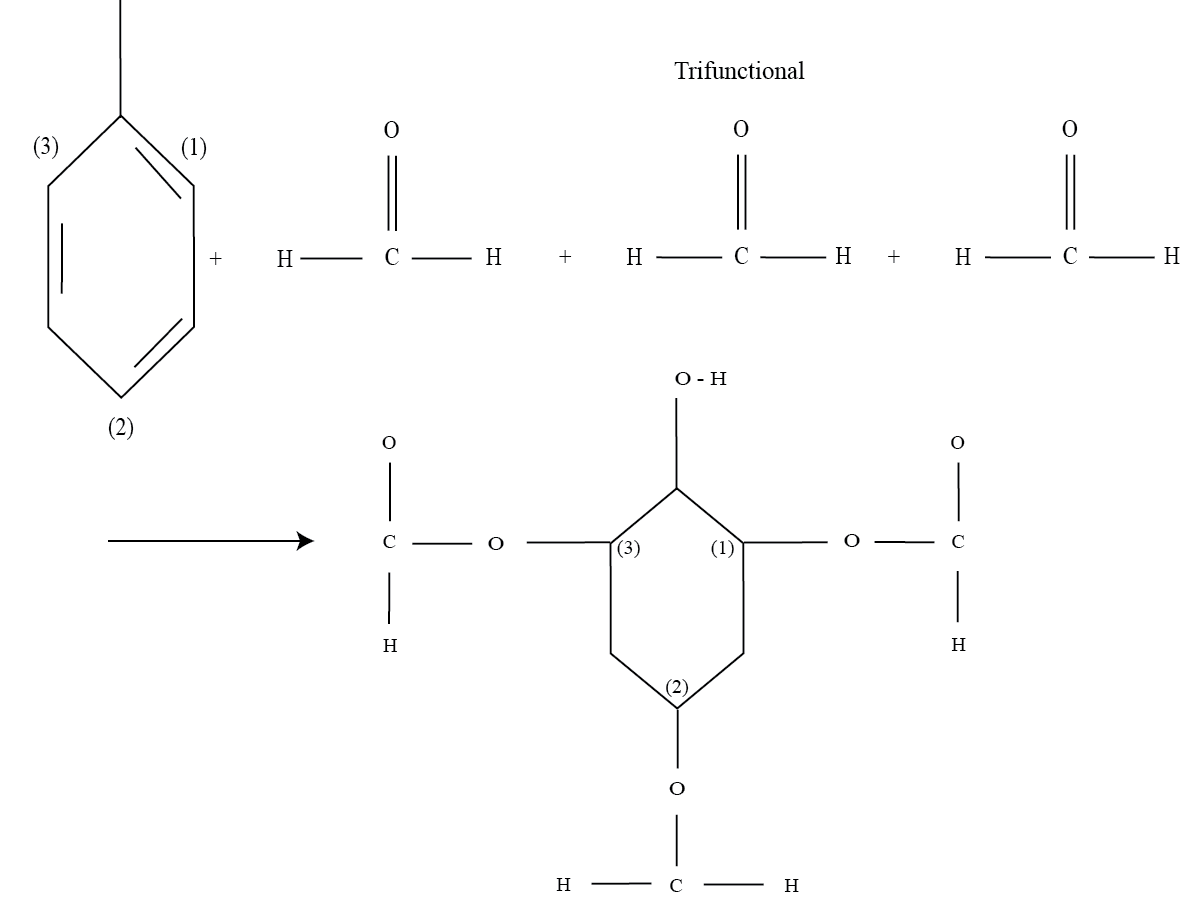
In the figure , the three formaldehyde molecules contain three reactive groups in the form of -OH groups. These reactive groups get attached to the positions 1, 2 and 3 in the phenol molecule.