Growth Mechanisms :
The growth mechanism of CNT on metal based catalysts and nonmetal based catalysts are discussed below.
i.
metal based catalysts
The growth mechanism of CNTs is yet to be fully established. In general it is proposed that hydrocarbons adsorb on metal particles and are catalytically decomposed. This results in carbon dissolving into particle. Upon supersaturation, carbon precipitates in tubular crystalline form. However, various alternative models have been proposed and appropriate description of growth depends on synthesis route and conditions used. Two most described models are root growth and tip growth [1]. The growth mechanisms are schematically explained in Fig. 4. For formation of CNTs the metal catalysts have to be dispersed well on the substrate forming nano clusters on the substrate surface. When there is a strong interaction between the catalyst clusters and substrate, the CNT grows by the root-growth mechanism. The pyrolysis of hydrocarbons produces the carbon atoms which are extremely mobile on metal surfaces and rapidly diffuse over and through the metal particles. The graphite precipitates around the catalyst particles and cylindrical structures are formed in a nested fashion from the catalyst particle, with the catalyst particle at the root. The growth of the nanotubes stops when the catalyst particles are completely covered with layers of carbon. The tip growth mechanism is observed when the interaction between metal catalyst clusters and substrate is weak. Due to the weak interaction, the catalyst particles are lifted off the substrate by the cylindrical structures formed below it. While, the carbon nanotube base remains anchored to the substrate, the tip carrying the catalyst particle grows toward the region of higher feed gas concentration. The tip growth stops when the catalyst particle is covered with carbon layers or when the supply of feed gas is cut off.
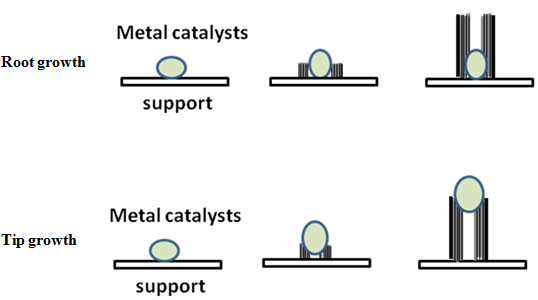
Fig. 4. Schmatics showing root growth and tip growth mechanism of CNTs
Synthesis of carbon nanotubes over Fe catalyst on aluminium is described by Emmenegger et al. [2] as follows. Initially iron nitrate layer on support transforms to crystalline Fe2O3 film during heating under nitrogen. After acetylene is introduced, Fe2O3 film is fragmented and smaller particles are formed. Fe2O3 crystals are reduced to inter-mediate oxides such as Fe3O4, FeO by hydrogen released from pyrolysis of acetylene. Finally metastable Fe3C is formed and growth of CNT occurs by tip growth mechanism.