Carbon nanotubes (CNT)
Materials with nano sized channels such as carbon nanotubes have received significant attention in recent years. In heterogeneous catalysis CNTs are being investigated as nano reactors, supports, active components and adsorbents. Other applications include electronic devices, gas and biosensors, nano-balance, scanning probe tips etc.
Carbon nanotubes were discovered in 1991 as minor byproduct during synthesis of fullerene which is an allotrope of carbon, in which the atoms are arranged in closed shells. Fullerenes consist of 20 hexagonal and 12 pentagonal rings as the basis of an icosahedral symmetry closed cage structure. The structure is shown in Fig 1.
Carbon nanotube structures consist of graphene cylinders closed at either end with capscontaining pentagonal rings. C70 is smallest nanotube. Nanotubes are formed by rolling up a graphene sheet into cylinder and capping each end with half of a fullerene molecule. Different wrapping results in different structures (Fig. 1) and electronic properties.
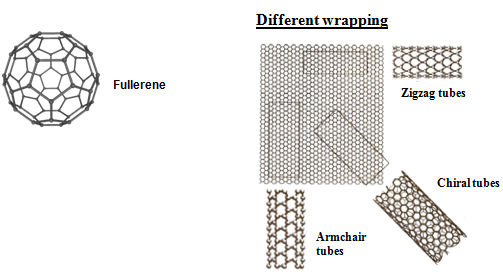
Fig. 1 : Structures of fullerene and carbon nanotube
In zigzag tubes some of the C-C bonds lies parallel to tube axis and in armchair tubes few C-C bonds lies perpendicular to tube axis. Chiral tubes have intermediate orientation.