Synthesis of CNT
There are two main methods for preparation of CNT
- Sublimation of graphite with subsequent desublimation
- Decomposition of carbon containing compounds
The most used method to prepare CNT is pyrolysis of hydrocarbon gases or vapors such as propane, butane, hexane, benzene, toluene etc. The method is also known as chemical vapor deposition (CVD) process.
This method involves condensation of carbon atoms generated from evaporation of solid carbon sources of graphite. The sublimation of the solid can be done using electric arc or laser ablation where the temperature reaches to 2500 -3500°C.
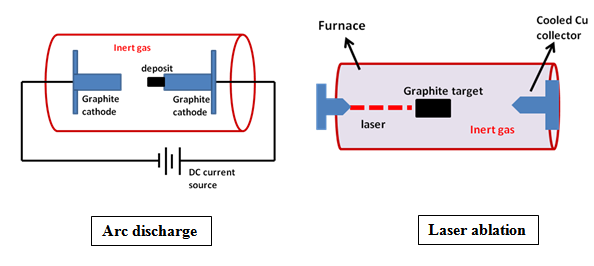
Fig. 2. Schematics for CNT formation by sublimation of graphite with subsequent desublimation.
The electric arc discharge method is one of the efficient techniques for synthesis of CNT. Typically, about 60 to70 wt% of the arc-synthesized soot is CNT. The rest of the soot comprises of fullerenes, amorphous carbon and catalyst nanoparticles. In electric arc discharge production of CNT two graphite rods are used and a current is passed continuously between the electrodes. The anode is drilled and filled with catalysts. The metal oxides (Ni, Co, Fe) are used as catalyst. In some cases the catalyst/graphite composite is used as electrode. The synthesis is performed in cooled chamber in presence of helium, argon or methane environment. During the arcing, the catalyst/graphite anode is evaporated and consumed with simultaneous carbon deposition around the cathode. The quality of CNT samples depends upon arc stability, current density and cooling of cathode. In laser ablation method the graphite target is subjected to laser and sublimated carbon is recollected. Inert gas atmosphere is maintained within the chamber.
Chemical Vapor Deposition ( CVD )
By chemical vapor deposition CNTs can be produced in large quantities. The process temperature can vary from 500 – 1300°C. The hydrocarbon precursors include CH4, C2H2, C6H6, alcohols etc.
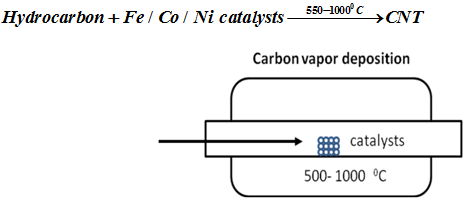
Fig. 3 . Schematics for carbon vapor deposition method
In CVD method there is initial dissociation of hydrocarbons followed by dissolution and saturation of C atoms in metal nanoparticles. Thereafter there is precipitation of carbon. Vapor-grown CNTs generally use metal catalyst particles. Fe, Co and Ni catalysts are mostly used for the catalytic growth of CNT. More recently, CNTs have also been grown from metal such as Au, Ag and Cu. Catalyst serves as nucleation sites and also promotes pyrolysis of hydrocarbons.