Metallocenes catalysts
Metallocenes are highly stereo specific catalysts having increasing applications. These catalysts consist of transitions metal (Zr, Ti or Hf) sandwiched between cyclopentadienyl rings to form a sterically hindered site. Typical structure is shown in Fig.3.
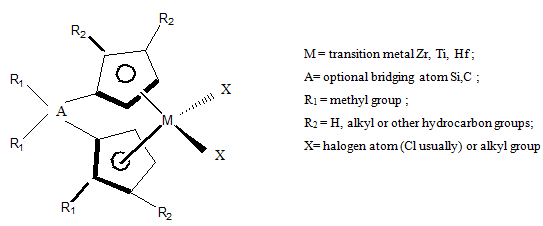
Fig. 3. Structure of typical metallocenes catalysts
These catalysts have high activity as well as stereoselectivity. The catalyst can produce either isotactic or syndiotactic polypropylene and are called single site catalyst. Polymers produced by metallocene catalysts have narrow molecular weight distribution. The main limitation of metallocene catalysts is their higher cost compared to conventional Ziegler – Natta catalyst. But due to higher activity and gradually decreasing price scenario metallocene based industrial polymerization process are rapidly growing.
Supported metal oxide catalyst
Most industrial polymerization catalysts are supported. Supported metal oxide catalysts include Cr, Mo, Co or Ni supported on alumina, silica, zirconia and activated carbon. They are used commercially for low pressure polymerization of alkene. The most active catalysts are Cr/SiO2, Zr/Al2O3 and Ti/MgO. These catalysts are observed to be active for ethylene polymerization but are less effective for propylene production because of low stereoregularity.
Chromium catalysts are extensively used in production of high density polyethylene HDPE. These catalysts are of two types; supported chromium oxide and organometallic compounds such as bis(arene)Cr0. Chromium oxides are supported on silica, alumina or titania. The supports affect the molecular weight distribution. Sometime mixed composition of inorganic and organic catalysts are also used.
The supported catalysts are rapidly poisoned, fouled or encapsulated by the polymer product. To maintain catalyst activity, the catalyst must constantly undergo fragmentation to expose new active catalytic sites.