b. Copolymer
Copolymers are prepared from two or more type of polymers. Different types of copolymers are obtained depending on the sequence of bonding of two different homochain polymers say A and B as shown in Fig. 2. Random copolymers have lower crystallinity and greater elasticity. In block polymer, blocks of one type of homopolymer structure are attached to blocks of another type of homopolymer. As a result in block polymer, desirable properties from each of the co-monomers are obtained. The styrene-butadiene thermoplastic elastomer is an example of block polymer while t he acrylonitrile-butadiene-styrene (ABS ) impact polymer is an example of network polymer.
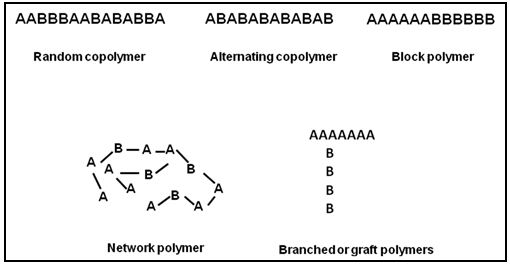
Fig. 2. Different types of copolymers that can be obtained from two homopolymers
A & B
Polymers can be produced by both heterogeneous and homogeneous catalytic processes though most industrial catalysts are heterogeneous. Catalysts are essential for initiation and/or control of the polymerization process. Polymerization processes are different from the other conventional catalytic processes in the fact that the catalysts are rarely recovered unchanged at the end of the reaction. The catalysts generally remain within the products as the separation cost is too high. Typically more than one ton of polymer per gram of catalyst is produced.
Polymerization can be done either by :
- Step growth (condensation) reaction or
- Chain growth (addition or insertion) reaction