Polymerization
Polymers are macromolecules obtained by bonding monomers which are small molecules consisting of unit structure of polymer that are repeated. Polymers with desirable properties of toughness, strength and elasticity have molecular weight in the range of 104 to 106 g/mol. Polymer can be classified in different ways:
1. Based on thermal behavior
- Thermoplastic: These are range of polymers that soften on heating.
- Thermoset: This class of polymers have rigid three dimensional structure and are not softened on heating.
- Elastomers: These polymers have low crystallinity and high flexibility.
2.Based on structure
- Homochain polymer
These polymers are synthesized from single monomer such as alkene (propylene, vinylchloride, styrene) or dienes (butadienes). Different stereochemical arrangements of substitutional R group around the carbon chain as shown in Fig. 1 result in polymers with variation in properties. In isotactic arrangement substitutional R group lies on the same side of the carbon chain as shown in Fig.1. In syndiotactic form R group alternate on either side of the carbon chain. When there is random arrangement of the substitutional R group on carbon chain the arrangement is known as atactic. The stereoregularity of polymer has significant effect on the properties of polymers. Isotactic and syndiotactic are crystalline where as atactic polymer are amorphous. Polypropylene exists in these forms. Isotactic polypropylene polymer is semicrystalline material while atactic polypropylene has rubber like properties.
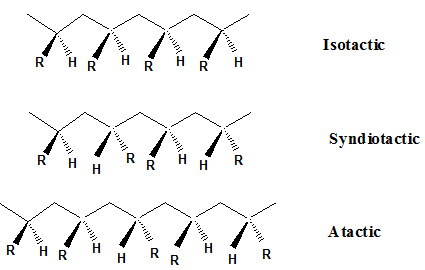
Fig. 1 . Different stereochemical arrangements of homochain polymers