4. Catalysis by Lewis acids
- Diels alder reactions
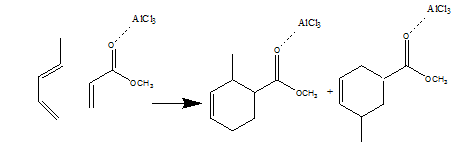
Reaction of diene with a mono-ene form cyclohexene derivative is shown below.
- Epoxidation
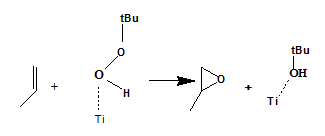
Epoxidation reaction is important reaction for producing organic intermediates. Alkenes can be transformed to epoxide by hyperoxides and catalysts. Catalysts are often titanium or molybdenum complex acting as Lewis acid.
5. Catalysis by Porphyrin complexes
Porphyrin complexes are used to catalyze epoxidation and hydroxylation reactions. The porphyrins are macrocyclic compound. The porphyrin molecule contains four pyrrole rings linked via methine bridges. The structure of porphyrin macro molecule is shown in Fig. 2. The porphyrin ring system is very stable and exhibits aromatic character. The porphyrin nucleus is a tetradentate ligand in which the space is available for a coordinating metal and has a maximum diameter of approximately 3.7 A0. When coordination occurs, two protons are removed from the pyrrole nitrogen atoms, leaving two negative charges. Various metals such as Na, K, Li Co, Ni, Cu, Fe, Mn form complexes.
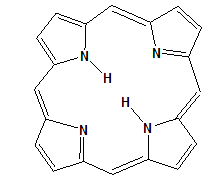
Fig 2. Structure of porphyrin macro molecule
Epoxidation
The Ru(II) porphyrin complexes is used as epoxidation catalyst of olefins. Ruthenium(II) carbonyl tetraphenylporphyrins catalyze epoxidation of olefins in presence of metachloroperbenzoic acid as oxidants.
Hydroxylation
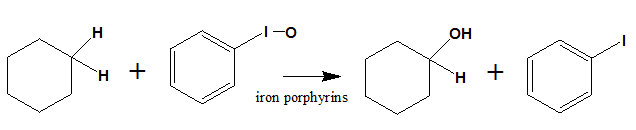
The hydroxylation of unactivated alkanes can be done in presence of iodosylbenzene and iron porphyrins catalyst. The oxidation of cyclohexane in presence of iodosylbenzene with chloro(5,10,15,20-tetra-o-tolylporphyrinato)iron(III) [Fe(TTP)CI] produce cyclohexanol and cyclohexanone as show below.
Text reference
- Piet W.N.M. van Leeuwen, Homogeneous catalysis: Understanding the Art, Springer, 2004
- Piet W.N.M. van Leeuwen, and John C. Chadwick, Homogeneous catalysis: Activity-stability –deactivation, Wiley, VCH, 2011
- H. Bartholomew and R. J. Farrauto, Fundamentals of Industrial catalytic Processes, Wiley, VCH, 2006
- M. Biesaga, K. Pyrzynska, M. Trojanowicz, Porphyrins in analytical chemistry: A review, Talanta 51 (2000) 209–224