Examples
- Acid catalyzed condensation
- Acid catalyzed condensation of phenol and acetone to bisphenol which is an important intermediate in manufacture of epoxy resin and polycarbonates.
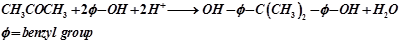
- Acid catalyzed synthesis of ethyl acetate ester from ethanol and acetic acid.
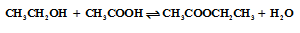
- Acid catalyzed condensation of phenol and acetone to bisphenol which is an important intermediate in manufacture of epoxy resin and polycarbonates.
- Acid catalyzed dehydration of ethyl alcohol to ethylene
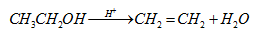
- Hydrolysis of ester
- Hydrolyses of carboxylic esters to form the parent carboxylic acid and an alcohol.
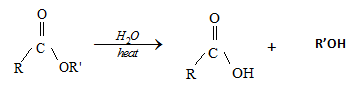
- Hydrolyses of carboxylic esters to form the parent carboxylic acid and an alcohol.
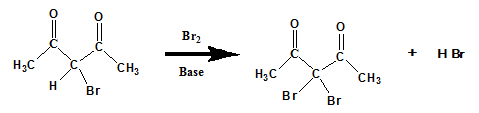
- Acid or base catalyzed halogenation
Ketones can be halogenated in the presence of acid or base and X2 (X= Cl, Br).
2. Catalysis by Metal ion
Metal ions can act as catalysts. Metal ions function in different ways :
- Metal ions can act as “super acid”. It introduces positive charge into the substrate, making it more susceptible toward nucleophilic attack.
- Metal ions can also act as template. Metal ions are able to coordinate to more than 2 ligands and thereby bring the molecules together.
- Metal ions can act as redox catalyst. Many metal ions can accept or donate electrons by changing their oxidation state and thereby participate in redox reaction.
Examples
- Catalysis by Cu2+ ions
Cu2+ ions are very effective catalysts for the hydrolysis of α-amino acid esters.
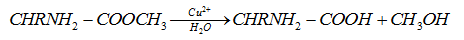
- Catalysis by Mg2+ ions
Hydrolysis of phosphate esters is catalyzed by metal ions, usually Mg2+.
![]()