3. Catalysis by Organometallic complexes
Presently, organometallic catalysts play major role in homogeneous catalysis. Organometallic complex consist of a central transition metal ion bonded to organic ligands such as R2C=CR2, RCO, R3P, R3N, CO etc. Catalysis occurs through dissociation of ligands followed by co-ordination of reactant molecule to the metal ion. The transition metal ions react through exchange of d electrons. Organometallic complexes usually have octahedral or tetrahedral geometry. Reactions catalyzed by organometallic complexes include hydrogenation, hydroformylation, carbonylation and decarbonylation, hydrocarbon rearrangement, partial oxidations etc.
Effect of ligands
The nature of surrounding ligands is very important in organometallic catalysis and known as ligand effect. The product distribution depends on the ligand environment around the metal center. Using the same metal center, different products can be obtained with the same substrate when associated ligands are changed around the metal center.
Ligand types
Phosphine based ligands ( PR3 ; where R= t-Bu, n-Bu, Ph, CH3O, CF3CH2O, Cl, CF3 ) are most widely used. The alkyl phosphines are strong bases and are σ donor ligands while the organophosphites, P(OR)3 , are strong π acceptors and form stable complexes with electron rich transition metal by accepting π electrons.
Other ligands that are used discussed below :
-
Hydrocarbyl group : Cyclopentadieneyl ligands (Cp) are associated with metals such as Ti, Zr, Hf. The Cp2TiCl2 catalyst is used in ethylene polymerization. Ruthenium complexes containing aromatics ligands are used for hydrogen transfer reactions such as transfer of hydrogen from alcohol to ketone producing another alcohol.
-
Alkoxide, imides and imido are used as anionic ligands in zirconium and titanium catalysts for the polymerization of alkene. Mostly used in combination with cyclopentadienyl ligands.
-
Nitrogen ligands include pyridine and imidazole ligands. They are more stable than phosphine based ligands. Enzyme catalysts contains mostly nitrogen ligands in the form of imidazoles or porphyrins binding to metals such as copper or iron. Some other nitrogen based ligands such as amido ligands, diimine ligand are shown in Fig 1.
- Some other ligands such as phosphine with nitrogen substituents, carbon based ligands are also shown in Fig. 1.
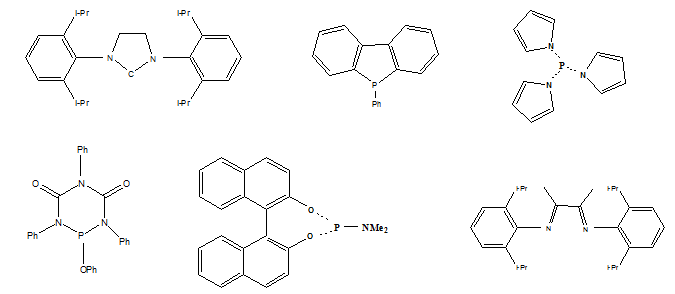
Fig. 1. Examples of phosphorous, nitrogen and carbon based ligands