- State the basic steps in surface area analysts. Which working equation is used and why.
- What different methods are available for measurement of pore volume and pore size distribution. Why this characterization is important for catalyst.
- How you can determine the crystal size and phase distribution of catalyst ?
- Why high dispersion of active metal is important in catalysis?
- How metal dispersion is determined?
- For which catalyst you can use the temperature programmed reduction technique ?
- Name the parameters affecting the TPR profiles.
- State the techniques used to determine the acidic and basic sites on catalysts surface.
- Write the characterization techniques that you will use to find the following characteristics of the V2O5 /Al2O3 catalyst :
(i) Reducibility of vanadia (ii) Meso pore size distribution (iii) Acidic strength of catalyst (iv) Surface topography of catalyst (v) Surface species of vanadia on support (vi) Phase distribution
- How does IR and Raman spectroscopy complement each other ?
- What is the finger print region in IR spectra?
- Draw the interferometer used in FT-IR depicting its basic function.
- State the difference between TGA and DTA. State advantage and limitation of each technique.
- Define magnification and resolution of a microscope.
- What are the advantages of electron microscope over optical microscope?
- Write the functions of
- Intermediate lens in TEM
- Objective lens in SEM
- State the basic difference in SEM and TEM techniques.
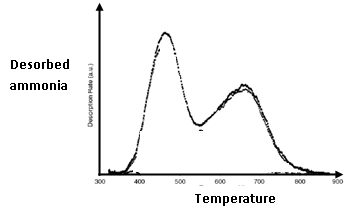
- For an oxide material the TPD profile of ammonia desorption as a function of temperature is given below. What informations can you derive from it ?