Then if ‘r' is the actual pore radius and the thickness of the adsorbed film is t ( Fig 4) then
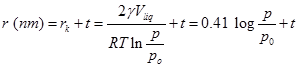 ---------------------------------(3)
---------------------------------(3)
Pore size distribution can be obtained by the analysis of either adsorption or desorption isotherm branches. Kelvin equations as described is applied to the desorption branch of hysteresis loop as it is more appropriates to assign wetting angle to a pore filled with liquid that possess a well defined meniscus. If ‘ h ' is the effective height of a monolayer then thickness of adsorbed layer ‘ t ' is given as 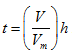 .
Here ‘V' is the volume of gas adsorbed and ‘Vm' is the volume of adsorbed monolayer.
When the packing of adsorbate is in hexagonal mode then for
nitrogen h = 3.6A0 and for cubic packing h= 4.3 A0 = 0.43 nm . The ‘t ' at a given relative pressure can also be calculated using Halsey equation
.
Here ‘V' is the volume of gas adsorbed and ‘Vm' is the volume of adsorbed monolayer.
When the packing of adsorbate is in hexagonal mode then for
nitrogen h = 3.6A0 and for cubic packing h= 4.3 A0 = 0.43 nm . The ‘t ' at a given relative pressure can also be calculated using Halsey equation
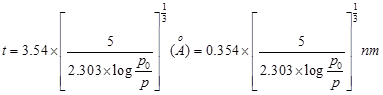 -----------------------------(4)
-----------------------------(4)
Pore size distribution
Most common method for determination of pore size distribution is BJH (Barrett-Joyner-Halenda) method. Assumptions are:
(i) Condensation occurs in pores when a critical relative pressure is reached corresponding to the Kelvin radius ‘rk'
(ii) When evaporation or condensation occurs, a multilayer of adsorbed film exist on the pore wall and this film has same depth/thickness as the adsorbed film on a non porous surface
(iii) Actual pore volume evaporation is composed of the volume evaporated out of the central core plus the volume desorbed from the film left on the pore walls
Steps in for determination of pore size distribution are as follows:
1. P/P0 and Vgas (STP, cm3/g) data obtained directly from isotherm
2. Then Kelvin radius ‘rk' is calculated from Kelvin equation using zero wetting angle for N2 from equation(2)
3. Then the film thickness t calculated from Halsey equation(4) at each P/P0
4. Then the pore radius rp calculated from equation (3)
5. Mean values of rk and rp in each decrement are calculated from successive entries
6. Change in film thickness Δt is calculated from difference of successive values of t
7. Then ΔVgas, that is the change in adsorbed volume between successive P/P0 values, is determined by subtracting successive values
8. Thereafter ΔVliq that is the volume of liquid corresponding to ΔVgas is calculated as follows
![]()
9. Then ΔtΣS is determined. This represents the volume change of the adsorbed film remaining on the walls of the pores from which the central core has previously evaporated. This volume is the product of the film area ∑S and the decrease in film depth Δt