Pore size
(a) By gas adsorption
Pore size and pore size distribution can be determined using Kelvin equation. Kelvin equation relates equilibrium vapor pressure (P) of a liquid contained in a capillary to equilibrium pressure of the same liquid over a free surface (Po) :
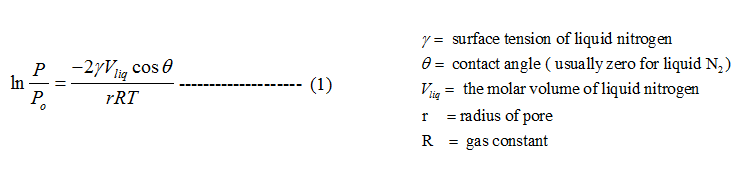
At any equilibrium pressure, P, pore of radius less than ‘r' will be filled with the condensed vapor. Application of Kelvin equation to all points of an isotherm at relative pressure greater than that corresponding to monolayer volume where capillary condensation begins to occur, will yield information concerning the volume of gas adsorbed in pores of different radii. For nitrogen as adsorbate and substituting values of various constants the Kelvin equation can be written as
![]()
or ![]() -------------------------------(2)
-------------------------------(2)
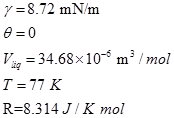
The ‘rk' is the radius into which condensation occurs at the required relative pressure. This radius is called Kelvin radius. However, Kelvin radius is not the actual pore radius since some adsorption has already occurred on the pore wall prior to condensation leaving a central core of radius rk. Conversely an adsorbed film remains on the wall when evaporation of the centre core takes place.
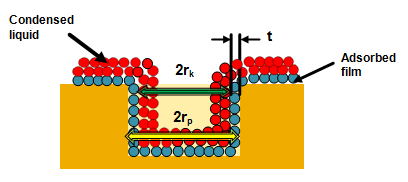
Fig. 3. Schematic showing Kelvin radius and actual pore radius during pore condensation