Multilayer adsorption theory
Langmuir equation is valid for adsorption less than monolayer coverage and therefore more suited for chemisorption studies as it involves only monolayer coverage. Physical adsorption is not limited to monolayer formation and multilayer adsorption can occur on subsequent layers. Hence Langmuir isotherm is not applicable to multilayer adsorption. Brunauer, Emmette and Teller derived an relationship between the volume of gas physically adsorbed and the partial pressure of the adsorbate which is valid for multilayer adsorption. This relationship is utilized for measuring the total surface area of an adsorbent.
Multilayer adsorption: BET equation derivation
Let be the surface area covered by 0,1,2,3……….i layers of adsorbed molecules. as shown in the Fig. 1. The total area of the adsorbent therefore, can be obtained by the summation of all the individual surface areas.
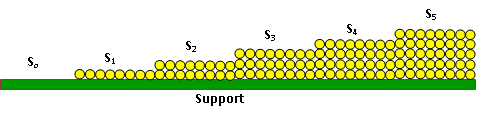
Fig.1. The representation of s0, s1, s2, s3, s4 and s5 surface areas covered by 0,1,2,3,4 and 5 layers of adsorbed molecules respectively.
Rate of adsorption on bare surface s0 is =
Rate of desorption from surface area s1 having the first layer =
Since at equilibrium s0 must remain constant, the rate of adsorption (or condensation) on bare surface is equal to the rate of desorption (evaporation) from first layer. Therefore,
or , where
heat of adsorption of first layer.
This is Langmuir equation for unimolecular adsorption. It is assumed that a, b and H are independent of the number of adsorbed molecules already present in the first layer.
Similarly, the rate of adsorption on s1= rate of desorption from s2.
Therefore,
Extending to layer si with ‘i' no of layers,
Total surface area , si = surface area with‘i' no of layers
Total volume adsorbed
or ,
----------------------------------------------------- [1]