(D) Sectioning: Initially, thick sections are cut with the help of razor blade to reach the tissue in the block. Afterwards, block is mounted on the microtome and ultra thin (10-100nm) sections are cut and floated onto the water placed within the boat. Cut sections are stacked on each other in a definite pattern known as “ribbon”. These ribbons are collected on electron microscope grid.
(E) Staining to visualize cell structure: Incubate the grid in uranyl acetate for 2hrs and subsequently in lead citrate for additional 2hr. Uranyl acetate staining will allow to observe the cellular structure and to study the changes in the cellular or sub-cellular morphology.
(F) Immunogold labeling:
- Block the grid sample with 5% BSA for 45 mins at room temp.
- Incubate the grid in the primary antibody for 1hr at room temperature or overnight at 40C in humified petridish.
[it is adviable to centrifuge primary antibody on full speed for 5mins to remove aggregated antibod].
- Dip the grid in PBS drop 3 times.
- Dip the grid in PBS containing tween 20 drop for 6times.
- Incubate the grid in the secondary antibody (antibody coupled to 10nm gold nanoparticle] for 1hr at room temperature or overnight at 40C in humified petridish.
[it is adviable to centrifuge secondary antibody on full speed for 5mins to remove aggregated antibod].
- Dip the grid in drop of PBS containing tween-20 for 6 times.
- Dip the grid in PBS drop for 3times.
- Finally stain the grid again with uranyl acetate for 1min and lead acetate for 30sec.
Observation: A typical TEM image is given in the Figure 35.2. As you can observe that cell shows black dots which indicate the presence of secondary antibody.
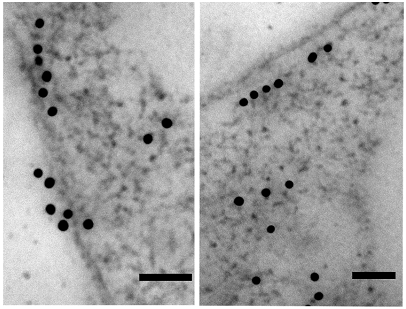
Figure 35.1: A typical image of eukaryotic cell observed with TEM. Gold particle, 10 nm; Bar, 50 nm