In chromatography, the stationary and mobile phases are not discrete but continuous. Paper chromatography, where filter paper was used as the stationary phase, was the very first chromatographic method. The paper chromatography technique developed with time and other stationary phases also emerged. Introduction of silica gel as a stationary phase led to the development of thin layer chromatography and subsequent functionalization of silica resulted in the development of various other chromatographic methods.
Today, most chromatographic methods use a column where stationary phase is a hydrated gel while the solvent/buffer flowing through the column makes the mobile phase. The gel is made up of small, homogeneous beads. Let us have a look at some important terminology we would be using in subsequent lectures:
Column : Column of the gel, not the actual vessel holding the gel
Column volume (Vt) : Total volume of the column; for a cylindrical column of radius, r and height, h, the volume is given by:
![]()
Void volume (V0) : The volume of the space occupied by the solvent outside the gel beads.
Included volume (Vi) : Volume of the solvent present inside gel beads.
The unhydrated gel matrix usually occupies ~1% volume of that of the hydrated one. Vt can therefore be approximated as:
![]() (Figure 20.3)
(Figure 20.3)
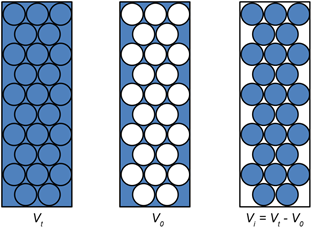
Figure 20.3: Terminology of the column volumes