Near-UV radiation
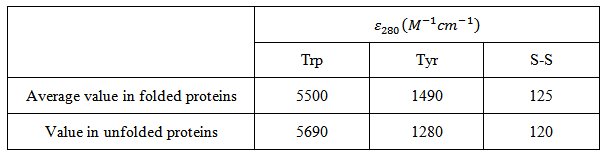
Aromatic amino acids, tryptophan, tyrosine, and phenylalanine and the disulfide linkage constitute the chromophores that absorb in the near UV region. Absorption of near UV radiation by proteins is usually monitored at 280 nm due to very high absorption by Trp and Tyr at this wavelength. Table 4.1 shows the molar absorption coefficient of the protein chromophores that absorb the light of 280 nm.
Table 4.1 Molar absorption coefficients of protein chromophores at 280 nm |
where, ε280 is the molar absorption coefficient at 280 nm.
It is therefore straightforward to calculate the molar absorption coefficient of a folded protein if its amino acid sequence or composition is known:
|
|
(4.4) |
For short peptides that are usually unfolded in water, the molar absorption coefficients can be calculated using the following equation:
|
|
(4.5) |
Far-UV radiation
The proteins and peptides that lack aromatic residues and disulfide linkage do not absorb the near UV radiation. The concentration of such proteins and peptides can be estimated using far UV radiation. Peptide bond is the major chromophore in the far UV region with a strong absorption band around 190 nm (π → π* transition) and a weak band around 220 nm (n → π* transition). As oxygen strongly absorbs 190 nm radiation, it is convenient to measure absorption
at 205 nm where molar absorption coefficient of peptide bond is roughly half of that at 190 nm.
A 1 mg/ml solution of most proteins would have an extinction coefficient of ~30 – 35 at 205 nm. This means that the result obtained can have more than 15% error. An empirical formula, proposed by Scopes [1] provides the ![]() within ± 2%:
within ± 2%:
|
|
(4.6) |
Alternatively, the concentration can be estimated using Wadell's method[2] that relies on the absorbance at 215 and 225 nm:
|
|
(4.7) |