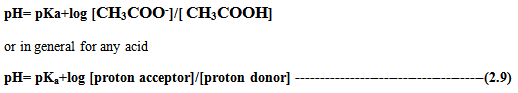The equation 2.9 is known as Henderson-Hasselbalch equation and it relates molar ratio of proton donor/acceptor with pKa at a given pH. It is also used to calculate the amount of proton donor, acceptor needed to prepare the solution of given pH, if pKa is given.
Buffer: The aqueous solution of weak acid or base and conjugated strong base (salt) and it resists to the change in pH on addition of small amount of acid or base. In biological system and in the in-vitro reactions are performed in buffer to keep the pH constant during the process. The degree of buffer solution to resist towards change in pH is calculated as buffering capacity , β. It is directly related to the pKa of the particular acid used in the buffer and buffering capacity is maximum at pKa and a safe range to use any buffer is pKa ± 1 unit.
 |
Role of buffer in biological system: each and individual organism keeps a constant pH to maintain homeostasis and growth. A number of biological buffers (including proteins) are known to maintain a constant pH within different tissues of human body. Few Buffers are specific to the organ or tissue where as many buffers are universally present. Biological reactions as well as macromolecules are sensitive to the change in pH and therefore buffer plays vital role for their optimal activity. |
Few examples of role of pH in controlling biological processes:
1. Enzymes are proteinous in nature and madeup of amino acids with ionizable side chains such as histidine. In addition, active site of enzyme has amino acid residues and a particular ionization state is important for substrate binding, formation of catalytic intermediates and release of products. For example, Pepsin is a serine protease present in stomatch and has a optimum pH of 1.5 where as trypsin has a pH optimum of 7.4.
2. In many pathological condition such as diabetes, body utilizes stored fat as an alternate energy source. The similar condition exist in the case of starvation or fasting and under these conditions a large amount of acid (β-hydroxybutyric acid) from fat is generated leading to the lowering of blood pH to cause “acidosis”. It disturbs activity of several enzymes present in blood and ultimately lead to headache, nausea and convulsions.
3. Blood pH is maintained by bicarbonate buffer system and play vital role in respiration.
4. Bacterial growth in mouth causes disturbance in mouth pH (acidity) and as a result it affects the mineral content of teeth resulting into the tooth decay.