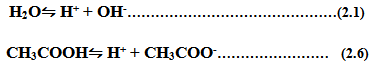![]()
Hence ability of any acid to dissociate to give H+ ion or a base to give OH- ion define the acid or alkali as strong acid or weak acid.
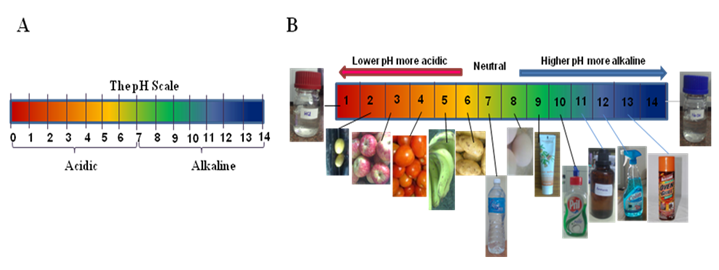
Figure 2.2: pH Scale and pH of different fluids. (A) pH Stripe, (B) pH in different biological fluids and daily use items. |
Handerson-Hasselbalch Equation- Titration is the method to determine the content of acid or base present in a solution. In a typical titration experiment, a fixed amount of acid is titrated with a solution of strong base, such as NaOH in the presence of pH indicator dye. The purpose of adding pH indicator dye is to monitor the change in pH during the course of titraton. A typical plot of change in pH against the amount of NaOH added for acetic acid is given in Figure 2.3. In the beginning, acetic acid is present majorly as CH3COOH but with the addition of NaOH, OH- will neutralize H+ to form H2O and promotes further dissociation of acetic acid to give H+ (Figure 2.3). At the mid point of where 0.5 equivalent NaOH has been added, solution has a equal concentration of CH3COOH (proton donor) and CH3COO- (proton acceptor). At the end of titration, all undissociated CH3COOH will be convereted into the CH3COO- .
During a titration experiment, two reversible equilibrium exists.