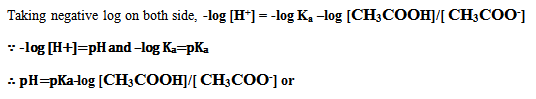![]()
pKa= -log Ka, Strong acid will have lower pKa. it is calculated by titration experiment and pH at the mid-point of the titration curve for acid or base (Figure 2.3).
| After rearranging, |
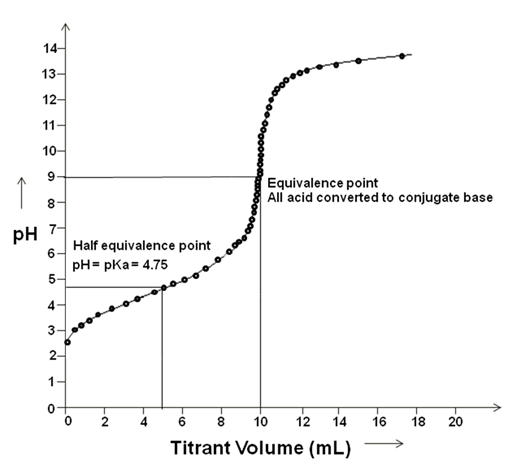
Figure 2.3: Titration Curve of acetic acid.
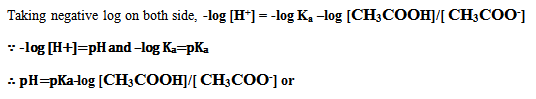
Module 1 : Introduction
Lecture 2: Water properties and its role in controlling biochemical reactions
![]()
pKa= -log Ka, Strong acid will have lower pKa. it is calculated by titration experiment and pH at the mid-point of the titration curve for acid or base (Figure 2.3).
| After rearranging, |

Figure 2.3: Titration Curve of acetic acid.