1. Cell size
Cell size is inversely correlated to the size of the external field needed to generate permeabilization. Consequently, optimization for each cell type is essential. Likewise, cell orientation matters for cells that are not spherical.
2. Temperature
It has been observed that plant membrane resealing is effectively temperature dependent and shows slow closure at low temperatures. For DNA transfer, it has been found that cooling at the time of permeabilization and subsequent heating in incubator increases transfer efficacy and cell viability.
3. Post-pulse manipulation
Cells are susceptible when in the permeabilized state, and it has been shown that waiting for 15min after electroporation in order to allow resealing before pipetting cells, increases cell viability.
4. Composition of electrodes and pulsing medium
For short pulses is needed for release of metal from the standard aluminium electrodes used in standard disposable cuvettes. Some authors advocate the use of low conductivity or more resistance media for DNA transfer in order to increase viability and increase transfection efficacy.
1. 3. Microinjection
The microinjection technique is a direct physical approach to inject DNA directly into the plant protoplasts or cells (specifically into the nucleus or cytoplasm) using fine tipped (0.5-1.0 µm diameter) capillary glass needle or micropipettes. Through microinjection technique, the desired gene introduce into large cells, such as oocytes, eggs, and the cells of early embryo (Figure 24.4).
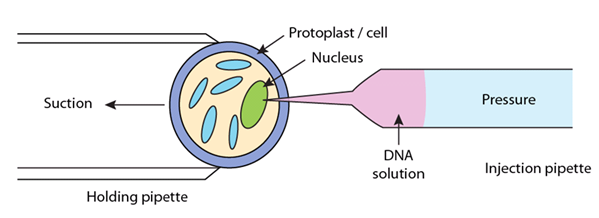
Figure 24.4: Microinjection
1.4. Liposome-mediated transformation
The idea of a method of direct plant transformation elaborated in the middle eighties was to introduce DNA into the cell by means of liposomes. Liposomes are microscopic spherical vesicles that form when phospholipids are hydrated. Liposomes are circular lipid molecules with an aqueous interior that can carry nucleic acids. Liposomes encapsulate the DNA fragments and then adhere to the cell membranes and fuse with them to transfer DNA fragments. Thus, the DNA enters the cell and then to the nucleus. Lipofection is a very efficient technique used to transfer genes in bacterial, animal and plant cells. They can be loaded with a great variety of molecules, including DNA. In the case of protoplasts, the transfection (lipofection) occurs through the membrane fusion and endocytosis. When pollen grains are transformed, liposomes are delivered inside through pores. The efficiency of bioactive-beads-mediated plant transformation was improved using DNA-lipofection complex as the entrapped genetic material instead of naked DNA used in the conventional method. Liposome-mediated transformation is far from routine, in spite of the low expense and equipment requirement. A probable reason is its laboriousness and low efficiency. Only few reports on the integration of genes introduced by means of liposomes followed by transgenic plant regeneration for tobacco and wheat have been published thus far.
1.5. Silicon carbide fiber mediated transformation (SCMT)
SCMT is one of the least complicated methods of plant transformation. Silicon carbide fibers are simply added to a suspension containing plant tissue (cell clusters, immature embryos, callus) and plasmid DNA, and then mixed in a vortex, or in other laboratory apparatus such as, commercial shakers, blenders etc. DNA-coated fibers penetrate the cell wall in the presence of small holes created in collisions between the plant cells and fibers. The most often used fibers in this procedure are single crystals of silica organic minerals like, siliconcarbide, which have an elongated shape, a length of 10–80 mm, and a diameter of 0.6 mm, and which show a high resistance to expandability. Fiber size, the parameters of vortexing, the shape of the vessels used, the plant material and the characteristics of the plant cells, especially the thickness of the cell wall are the factors depending on the efficiency of SCMT. There are several known examples of deriving transgenic forms, cell colonies or plants in maize, rice, Wheat, tobacco, Lolium multiflorum , Lolium perenne, Festuca arundinacea , and Agrostis stolonifera by SCMT.
SCMT is an easy, fast and inexpensive procedure. Therefore, it could be an attractive alternative method of plant transformation in particular situations, e.g. when a gene gun is not available and Agrobacterium -mediated transformation is difficult or not possible (as in the case of numerous monocots). The other advantages of the SCF-mediated method over other procedures include the ability to transform walled cells, thus, avoiding protoplast isolation .
The main disadvantages of this method are low transformation efficiency, damage to cells, thus, negatively influencing their further regeneration capability. Another disadvantage is that silicon fibers have similar properties to asbestos fibers and care must be taken when working with them as breathing the fibers can lead to serious sicknesses. Silicon carbide has some carcinogenic properties as well.