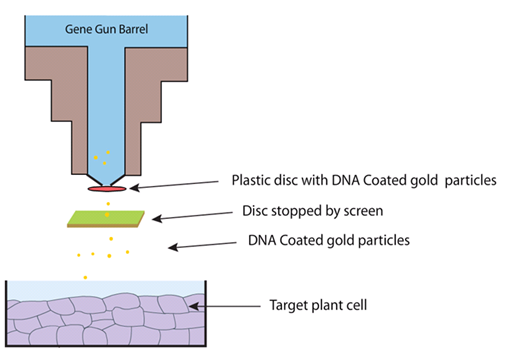
Figure 24.2: Diagrammatic illustration of gene transfer using Gene Gun method
The technique has many advantages and can be used to deliver DNA into virtually all the tissues, like immature and mature embryos, shoot-apical meristem, leaves, roots etc. Particle bombardment methods are also useful in the transformation of organelles, such as chloroplasts, which enables engineering of organelle-encoded herbicide or pesticide resistance in crop plants and to study photosynthetic processes.
Limitations to the particle bombardment method, compared to Agrobacterium-mediated transformation, include frequent incorporation of multiple copies of the transgene at a single insertion site, rearrangement of the inserted genes, and insertion of the transgene at multiple insertion sites. These multiple copies can be associated with silencing of the transgene in subsequent progeny. The target tissue may often get damaged due to lack of control of bombardment velocity.
1.2. Electroporation
Electroporation is another popular physical method for introducing new genes directly into the protoplasts. In this method, electric field is playing important role. Due to the electric field protoplast get temporarily permeable to DNA. In electroporation, plant cell protoplasts are kept in an ionic solution containing the vector DNA in a small chamber that has electrodes at opposite ends. A pulse of high voltage is applied to the electrode which makes the transient pores (ca. 30 nm) in the plasma membrane, allowing the DNA to diffuse into the cell (Figure 24.3). Immediately, the membrane reseals. If appropriately treated, the cells can regenerate cell wall, divide to form callus and, finally, regenerate complete plants in suitable medium. The critical part of the procedure is to determine conditions which produce pores that are sufficiently large and remain open long enough to allow for DNA diffusion. At the same time, the conditions should make pores that are temporary. With a 1 cm gap between the electrodes and protoplasts of 40-44µm diameter, 1-1.5 kVcm -2 of field strength for 10µs is required for efficient introduction of DNA. It was seen that presence of 13% PEG (added after DNA) during electroporation significantly raised the transformation frequency. The other factors which may improve the transformation frequency by electroporation are linearizing of plasmid, use of carrier DNA, and heat shock (45 ~ for 5 min) prior to addition of vector, and placing on ice after pulsing. Under optimal conditions transformation frequencies of up to 2% have been reported. Stably transformed cell lines and full plants of a number of cereals have been produced through electroporation.
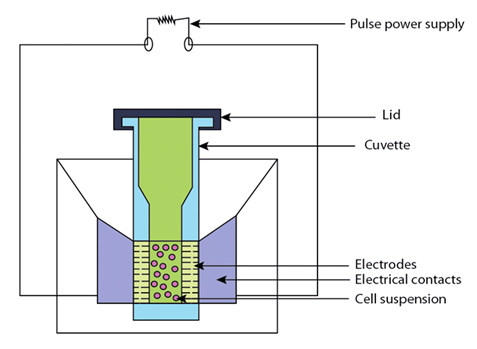
Figure 24.3: Electroporation
There are some parameters that can be considered when performing in vitro electroporation: