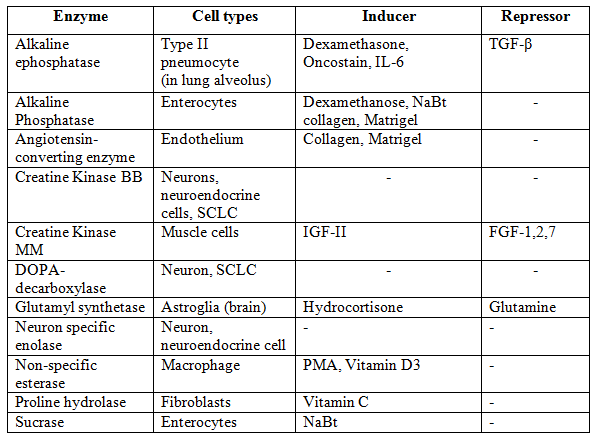
Table 1: Enzymatic markers used for cell line
e. Regulation: The level of expression of many differentiated products is under the regulatory control of environmental influences, such as nutrients, hormones, the matrix, and adjacent cell. Hence the measurement of specific lineage markers may require preincubation of the cells in, for example, a hormone such as hydrocortisone, specific growth factors, or growth of the cells on extracellular matrix of the correct type. f. Lineage fidelity: L ineage markers are more properly regarded as tissue or cell type markers, as they are often more characteristic of the function of the cell than its embryonic origin. |
5. Unique Markers: Unique markers include specific chromosomal aberrations (e.g., deletions, translocations, polysomy), major histocompatibility (MHC) group antigens (e.g., HLA in humans), which are highly polymorphic, and DNA fingerprinting or SLTR DNA profiling. Enzymic deficiencies, such as thymidine kinase deficiency (TK−) and drug resistance such as vinblastine resistance (usually coupled to the expression of the P-glycoprotein by one of the mdr genes that code for the efflux protein) are not truly unique, but they may be used to distinguish among cell lines from the same tissues but different donors.
6. Transformation: The transformation status forms a major element in cell line characterization and is dealt with separately.
- Cell Morphology: Observation of morphology is the simplest and most direct technique used to identify cells. Most of these are related to the plasticity of cellular morphology in response to different culture conditions. For example, epithelial cells growing in the center of a confluent sheet are usually regular, polygonal, and with a clearly defined birefringent edge, whereas the same cells growing at the edge of a patch may be more irregular and distended and, if transformed, may break away from the patch and become fibroblast-like in shape.
- Microscopy: The inverted microscope is one of the most important tools in the tissue culture laboratory, but it is often used incorrectly. As the thickness of the closed culture vessel makes observation difficult from above, because of the long working distance, the culture vessel is placed on the stage, illuminated from above, and observed from below. As the thickness of the wall of the culture vessel still limits the working distance, the maximum objective magnification is usually limited to 40X. The use of phase-contrast optics, where an annular light path is masked by a corresponding dark ring in the objective and only diffracted light is visible, enables unstained cells to be viewed with higher contrast than is available by normal illumination. Because this means that the intensity of the light is increased, an infrared filter should be incorporated for prolonged observation of cells.
It is useful to keep a set of photographs at different cell densities for each cell line, prepared shortly after acquisition and at intervals thereafter, as a record in case a morphological change is subsequently suspected. Photographs of cell lines in regular use should be displayed above the inverted microscope. Photographic records can be supplemented with photographs of stained preparations and digital output from DNA profiling and stored with the cell line record in a database or stored separately and linked to the cell line database.