Important Properties of Compressible Flows
The simple definition of compressible flow is the variable density flows. In general, the density of gases can vary either by changes in pressure and temperature. In fact, all the high speed flows are associated with significant pressure changes. So, let us recall the following fluid properties important for compressible flows;
Bulk modulus ![]() : It is the property of that fluid that represents the variation of density
: It is the property of that fluid that represents the variation of density ![]() with pressure
with pressure ![]() at constant temperature
at constant temperature ![]() . Mathematically, it is represented as,
. Mathematically, it is represented as,
(4.1.8) |
In terms of finite changes, it is approximated as,
(4.1.9) |
Coefficient of volume expansion ![]() : It is the property of that fluid that represents the variation of density with temperature at constant pressure. Mathematically, it is represented as,
: It is the property of that fluid that represents the variation of density with temperature at constant pressure. Mathematically, it is represented as,
(4.1.10) |
In terms of finite changes, it is approximated as,
(4.1.11) |
Compressibility ![]() : It is defined as the fractional change in the density of the fluid element per unit change in pressure. One can write the expression for
: It is defined as the fractional change in the density of the fluid element per unit change in pressure. One can write the expression for ![]() as follows;
as follows;
(4.1.12) |
In order to be more precise, the compression process for a gas involves increase in temperature depending on the amount of heat added or taken away from the gas. If the temperature of the gas remains constant, the definition is refined as isothermal compressibility ![]() . On the other hand, when no heat is added/taken away from the gases and in the absence of any dissipative mechanisms, the compression takes place isentropically. It is then, called as isentropic compressibility
. On the other hand, when no heat is added/taken away from the gases and in the absence of any dissipative mechanisms, the compression takes place isentropically. It is then, called as isentropic compressibility ![]() .
.
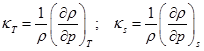 |
(4.1.13) |
Being the property of a fluid, the gases have high values of compressibility ![]() while liquids have low values of compressibility much less than that of gases
while liquids have low values of compressibility much less than that of gases ![]() . From the basic definition (Eq. 4.1.12), it is seen that whenever a fluid experiences a change in pressure
. From the basic definition (Eq. 4.1.12), it is seen that whenever a fluid experiences a change in pressure ![]() , there will be a corresponding change in
, there will be a corresponding change in ![]() . Normally, high speed flows involve large pressure gradient. For a given change in
. Normally, high speed flows involve large pressure gradient. For a given change in ![]() , the resulting change in density will be small for liquids (low values of k) and more for gases (high values of k). Therefore, for the flow of liquids, the relative large pressure gradients can create much high velocities without much change in densities. Thus, the liquids are treated to be incompressible. On the other hand, for the flow of gases, the moderate to strong pressure gradient leads to substantial changes in the density (Eq.4.1.12) and at the same time, it can create large velocity changes. Such flows are defined as compressible flows where the density is a variable property and the fractional change in density
, the resulting change in density will be small for liquids (low values of k) and more for gases (high values of k). Therefore, for the flow of liquids, the relative large pressure gradients can create much high velocities without much change in densities. Thus, the liquids are treated to be incompressible. On the other hand, for the flow of gases, the moderate to strong pressure gradient leads to substantial changes in the density (Eq.4.1.12) and at the same time, it can create large velocity changes. Such flows are defined as compressible flows where the density is a variable property and the fractional change in density ![]() is too large to be ignored.
is too large to be ignored.