Here, M is the molecular weight of the gas, R is the gas constant that varies from gas to gas and ![]() is the universal gas constant. In a calorically perfect gas, the other important thermodynamic properties relations are written as follows;
is the universal gas constant. In a calorically perfect gas, the other important thermodynamic properties relations are written as follows;
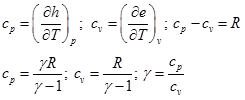 |
(4.1.2) |
In Eq. (4.1.2), the parameters are specific heat at constant pressure ![]() , specific heat at constant volume
, specific heat at constant volume ![]() , specific heat ratio
, specific heat ratio ![]() , specific enthalpy
, specific enthalpy ![]() and specific internal energy
and specific internal energy ![]() .
.
First law of thermodynamics : A system is a fixed mass of gas separated from the surroundings by a flexible boundary. The heat added ![]() and work done (w) on the system can cause change in energy. Since, the system is stationary, the change in internal energy. By definition of first law, we write,
and work done (w) on the system can cause change in energy. Since, the system is stationary, the change in internal energy. By definition of first law, we write,
(4.1.3) |
For a given de, there are infinite number of different ways by which heat can be added and work done on the system. Primarily, the three common types of processes are, adiabatic (no addition of heat), reversible (no dissipative phenomena) and isentropic (i.e. reversible and adiabatic).
Second law of thermodynamics : In order to ascertain the direction of a thermodynamic process, a new state variable is defined as ‘entropy ![]() '. The change in entropy during any incremental process
'. The change in entropy during any incremental process ![]() is equal to the actual heat added divided by the temperature
is equal to the actual heat added divided by the temperature ![]() , plus a contribution from the irreversible dissipative phenomena
, plus a contribution from the irreversible dissipative phenomena ![]() occurring within the system.
occurring within the system.
(4.1.4) |
Since, the dissipative phenomena always increases the entropy, it follows that
(4.1.5) |
Eqs. (4.1.4 & 4.1.5) are the different forms of second law of thermodynamics . In order to calculate the change in entropy of a thermodynamic process, two fundamental relations are used for a calorically perfect gas by combining both the laws of thermodynamics;
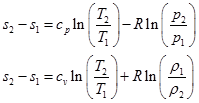 |
(4.1.6) |
An isentropic process is the one for which the entropy is constant and the process is reversible and adiabatic. The isentropic relation is given by the following relation;
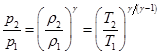 |
(4.1.7) |