3. ELISA plate coating: Add 100 µL diluted Capture Aantibody to each well. Incubate overnight at 4°C. Aspirate and wash 3 times with wash buffer.
4. Blocking : Add 200 µL Assay Diluent to each well. Incubate 1 hr RT. Aspirate and wash 3 times with wash buffer.
5. Add 100 µL standard TNF-α or sample to each well. Incubate it for 2 hr at RT. Aspirate the sample and wash the plate 5 times with wash buffer.
6. Detection: Add 100 µL Working Detector (Detection Ab + SAv-HRP) to each well. Incubate for 1hr at RT. Aspirate the detector solution and wash 7 times with wsh buffer. Add 100 µL Substrate Solution to each well and incubate 30 min RT in dark. Stop the reaction by adding 50 µL Stop Solution to each well. Read the ELISA plate at 450 nm and the at 570 nm (it is required to substract background absorbance).
Determination of the TNF-α level:- The mean mean absorbance of each set of sample and subtract the background absorbance from each mean. Draw a calibration by Plotting the standard TNF-α concentration against the absorbance. A typical standard TNF-α is given in the Figure 35.6. Use a regression analysis and draw the equation. Use this equation to determine the TNF concentration of the unknown sample.
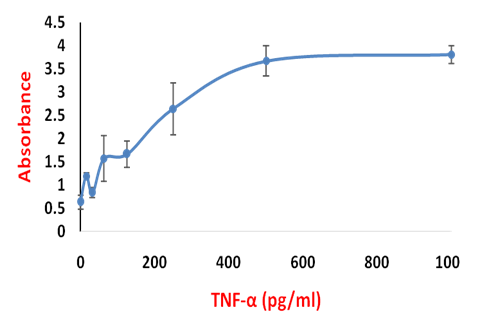
Figure 35.6: TNF-α standard Curve