| |
| Recap |
| In
this Lecture you have learnt the following |
| |
Summary |
| |
In this lecture, we have related a trajectory or a path on a PES to a collision process. Collisions can be classified into various categories. In a reactive collision, the trajectory moves from the reactant valley to the product valley by going through the region associated with the activated complex. In a non-reactive trajectory, the path returns to the starting reactant valley from an unfavourable configuration near the activated complex barrier. |
| |
The path along a classical trajectory can be investigated in time by solving the Newtons equations (or equivalently the Hamiltons equations) of motion. Although classical trajectories give an excellent picture of the passage of reactants to products, the correct picture is quantum mechanical wherein one obtains state to state rate constants, k { nR 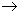 nP } wherein nR and nP represent the complete set of quantum numbers specifying the reactants and products. The macroscopic or the bulk rate constant can then be obtained by averaging over all the state to state rate constants by using an appropriate initial distribution over reactant states. nP } wherein nR and nP represent the complete set of quantum numbers specifying the reactants and products. The macroscopic or the bulk rate constant can then be obtained by averaging over all the state to state rate constants by using an appropriate initial distribution over reactant states. |
| |
|
| |
|
| |
|
| |
|
| |
|
| |
Congratulations, you have finished Lecture 35. To view the next lecture select it from the left hand side menu of the page |
|
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
| |
|