27.6) |
The dimerization of 2NO (g) 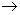 N2O2(g) was studied by noting the decrease in pressure of the reacting mixture as a function of time N2O2(g) was studied by noting the decrease in pressure of the reacting mixture as a function of time |
| |
|
| |
P total (in mm Hg) |
631 |
590 |
532 |
482 |
445 |
416 |
|
|
|
|
|
|
|
Time (in min) |
0 |
10 |
30 |
60 |
100 |
150 |
|
|
|
| |
Find the partial pressure of the monomer as a function of ime. If P0 in the initial pressure of NO and the partial pressure of NO has decreased by x at time t, then the pressure of remaining NO is P0 - x and the partial pressure of N2O2 is x/2 and the total pressure P total at t is P0 - x/2 or x = 2 (P0 - P total (t)) and the partial pressure of the monomer remaining is P0 - x = 2 P total - P0 Plot ln P0 / (2 P total - P0) vs time to see if the reaction is first order with respect to NO(g) |
|
| 27.7) |
Look up the detailed meanings of fluorescence, phosphoroscence, chemiluminiscence and give examples of molecules who have dominantly these properties. |
|
|
| 27.8) |
Try and find out how short pulses of the durations of a few pico seconds or a few femtoseconds are generated. |
| |