| |
them in the decreasing order of molar conductivity. |
| |
the reaction cell. Another part goes through a longer path (reflected by a mirror, passed through a fluorescent solution which converts the beam into continuous radiation in the visible range so that this can be used to analyse the products) so that it can reach the reaction vessel after a time delay to analyse the products. For a time delay of 10-7s, the additional path to be traveled by the second analysing beam is c * t = 2.988 * 1010cm/s * 10 -7 s = 2998cm. What is the smallest time delay that can be achieved in this set up? |
| |
reaction NaOH + CH3COOCH3 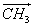 COONa + H2O The initial concentrations of both the reactants were 0.004 mol / lit. The molar conductivity COONa + H2O The initial concentrations of both the reactants were 0.004 mol / lit. The molar conductivity 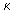 at different times was measured as follows at different times was measured as follows |
| |
Time (in minutes) |
1000 * 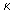 (in ohm –1 cm –1) (in ohm –1 cm –1) |
0 |
1.24 |
10 |
1.04 |
20 |
0.92 |
30 |
0.84 |
40 |
0.78 |
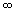 (very large times) (very large times)
|
0.44 |
|
| |
Initially the conductivity is mainly due to OH- and at the end, the conductivity is mainly due to CHCOO- ions. Find the concentrations of OH - and CH3COO - as a function of time. Determine the reaction order with respect to OH - by plotting log [ a/ (a-x) ] vs time. |