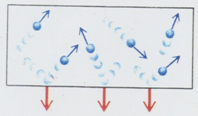
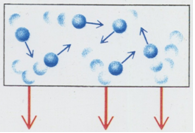
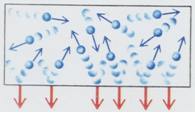
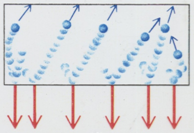
Some important features on pressure of an ideal gas
- Collisions of molecules with the wall lead to impulses. Their average
effect over a time interval is nothing but the pressure exerted by the gas.
- If mass of each molecule of the gas is doubled, the impulses are twice
as large. Thus pressure is proportional to molecular mass.
- If the gas density is doubled the number of collisions also become doubled.
Thus pressure is proportional to the gas density.
- If the average molecular speed is doubled, there are twice as many collisions
over the time interval and each impulses twice as large. Hence pressure
is proportional to square of molecular speed.