Enthalpy of Formation
Let us consider the steady state steady flow combustion of carbon and oxygen to from CO2 (Fig.3.3). Let the carbon and oxygen each enter the control volume at 25° C and 1 atm. Pressure, and the heat transfer be such that the product CO2 leaves at 25° C, 1 atm Pressure. The measured value of heat transfer is -393,522 kJ/kgmolof CO2 formed. If HR and HP refer to the total enthalpy of the reactants and products respectively, then the first law applied to the reaction C + O2 → CO2 gives
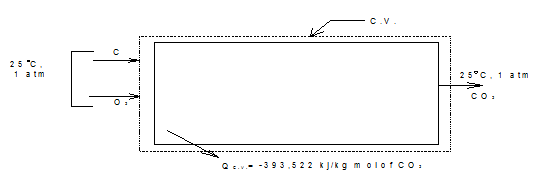
Fig.3.3 Enthalpy of formation
For all the reactants and products in a reaction, the equation may be written as
|
(3.93) |
where R and P refer to the reactants and products respectively. The enthalpy of all the elements at the standard reference state of 25° C, 1 atm is assigned the value of zero. In the carbon-oxygen reaction, HR = 0. Hence, the energy equation gives
QC.V = HP = -393,522 kJ/kg mol |
(3.94) |
This is known as the enthalpy of formation of CO2 at 25° C, 1 atm., and designated by the symbol, ![]() . In most cases, however, the reactants and products are not at 25° C, 1 atm., therefore, the change of enthalpy (in case of constant pressure or S.S.S.F. process) between 25° C, 1 atm.and the given state must be known. Thus the enthalpy at any temperature and pressure,
. In most cases, however, the reactants and products are not at 25° C, 1 atm., therefore, the change of enthalpy (in case of constant pressure or S.S.S.F. process) between 25° C, 1 atm.and the given state must be known. Thus the enthalpy at any temperature and pressure,![]() is
is
|
(3.95) |
For convenience, the subscripts are usually dropped, and
|
(3.96) |
where![]() represents the difference in enthalpy between any given state and the enthalpy at 298.15K, 1atm. Table 3.4 gives the values of the enthalpy of formation of a number of substances in kJ/kgmol.
represents the difference in enthalpy between any given state and the enthalpy at 298.15K, 1atm. Table 3.4 gives the values of the enthalpy of formation of a number of substances in kJ/kgmol.