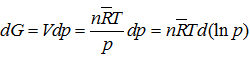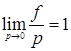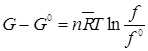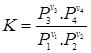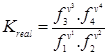Fugacity and Activity
The differential of Gibbs function of an ideal gas undergoing an isothermal process is
|
(3.84) |
Analogously, the differential of Gibbs function for a real gas is
|
(3.85) |
where ƒ is called the fugacity, first used by Lewis. The value of fugacity approaches the value of pressure, as the letter tends to zero, i.e., when ideal gas conditions apply. Therefore,
|
(3.86) |
For an ideal gas ƒ = p. Fugacity has the same dimension as pressure. Integrating Eq. (3.85)
|
(3.87) |
where G0 and ƒ0 ƒrefer to the reference state when p0 = 1 atm. The ratio ƒ/ƒ0is called the activity.
Therefore,
|
(3.88) |
For ideal gases, the equilibrium constant is given by
|
(3.89) |
For real gases
|
(3.90) |
Similarly, it can show that
|
(3.91) |
and
|
(3.92) |