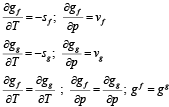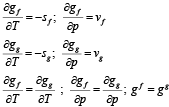The second order phase transition occurs at the critical point where saturated liquid flashes into vapor. There is no distinction of phases at triple point. However, there is distinct variation of the value of specific heat Cp. Following are the assumptions for second order phase transition
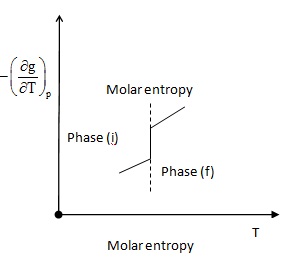
- There are no changes of entropy and specific volume
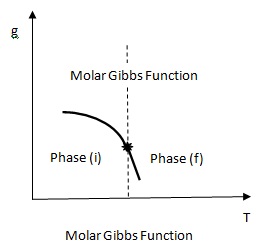
- Molar Gibbs function g is continuous
- First order derivative of g is continuous
|
|
Fig 2.11(a) |
Fig 2.11(b) |
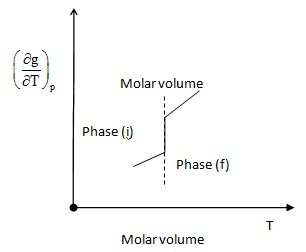
Fig 2.12
|
(2.25) |
|
|
(2.26) |
- Second order derivatives of Gibbs function g changes discontinuously.
|
(2.27) |
At critical point the latent heat of vaporization, l is zero.
(Remember: Fluid containing one component exhibits a single critical point.)