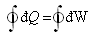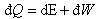About the first law of thermodynamics
Simple and most direct statement of the first law of thermodynamics is that energy is conserved. Energy can neither be created nor destroyed.
For a cyclic process involving heat and work First Law of Thermodynamics
is given by
For a thermodynamic process with heat and work interaction the expression
is given by
where E, Q, and W are the energy stored in the system, heat and work interacting at the system boundary, respectively.
- First Law gives a quantative measure of energy
- It can not distinguish the different types of energy
- It fails to indicate the direction of flow of energy
- It can not indicate whether the process is cyclic or not.
- First law applied to a process gives rise to a property (internal energy, U for a closed system and enthalpy, h for a open system)
From the definition of first law, it is obvious that energy is always conserved. First law efficiency indicates conversion of energy in a cycle as given by Eq. (1.1)
|
1.1 |
The net output can be work or heat depending upon the application. In case of a heat engine, the net output is in the form of work whereas in case of a solar water heating system it can be the useful heat. The difference between the net output and input is the energy unused. It is always the need from the first law of the thermodynamics to reduce the energy waste from the system. First law efficiency can be improved to limited extent with heavy insulation to the system.