| Polarization in Insulating Materials |
| |
| Polarization in insulating materials is basically a phenomenon of interaction between the applied external electric field and the inherent charge carriers, the atoms, ions or molecules present in the dielectric. Not only the applied electric field gives rise to the polarization, but in turn, polarization modifies the microscopic field within the dielectric. Thus, it is a process of a reversible displacement between the positions of positive and negative charges in the molecular structure caused by the applied electric field. The interaction takes place by a force, exercised on the basic structural elements (charge carriers) of the dielectric. |
The extent of polarization is analytically described by the relative permittivity εr . Consider a uniform field electrode system in vacuum applied an electric field E. The electric flux density 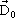 is given by, is given by, |
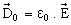 (16.3) (16.3) |
For the same electrode and the magnitude of the applied electric field, when the vacuum is replaced by an insulating material (solid, liquid or gas), the electric flux density in the dielectric 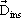 , is increased, given by, , is increased, given by, |
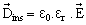 (16.4) (16.4) |
The increase in electric flux density, ' '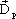 ' is caused by polarization in the dielectric. ' is caused by polarization in the dielectric. 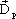 , therefore, represents the material property and is given as: , therefore, represents the material property and is given as: |
|
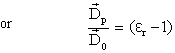 (16.5) (16.5) |
The quotient Dp / Do is known as dielectric susceptibility or polarization capacity of a dielectric. It is also denoted by χe |
Most of the gaseous dielectrics have their εr nearly equal to one. Hence polarization is not an important phenomenon in gases, but in liquid and solid dielectrics it plays an important role determining the conductivity and the losses. Different types of polarization mechanisms are described under the following three main categories.
|
Displacement Polarization
|
Within a molecular bond, the positive and negative charges of individual molecule or atom are rendered to oscillate in synchronism under the influence of an applied electric field. Similar oscillations also take place between the nucleus and the electron shell of an atom, building dipoles. The displacement caused by the oscillations is proportional to the applied electric field and on removing the electric field the atoms return back to their original state. Where only this type of polarization mechanism is present, the dielectric materials are described to be 'nonpolar' and these have no dipole moment. Classic examples of such dielectrics are the gases.
|
Space Charge or Boundary Surface Polarization
|
Some heterogeneous dielectrics, for example partially crystalline insulating materials, have the positive and negative ions as free charge carriers. Without an external field, the dielectric is in neutral condition and the positive and negative charges neutralise each other. However, under the effect of an external electric field, the charge carriers in the dielectric move towards opposite polarity electrode surfaces, giving rise to a macroscopic dipole. Like the mechanism described in previous case, in this care too on removing the applied electric field, the dielectric returns to its original state, hence it is known as a type of displacement polarization. |
Dielectrics having this type of polarization mechanism are also described as nonpolar, that is, these do not build a dipole without an external field. The boundary surface polarization is commonly present in heterogeneous insulating materials, such as, impregnated paper insulations, hard pressed boards, taped insulations used in electrical machines and even at voids in solid homogeneous dielectrics. Such materials normally have a low value of εr .
|
Orientation Polarization
|
Orientation polarization is the main polarization mechanism in liquid dielectrics. Usually, the polarization depends upon the applied electric field intensity. However, in some materials a permanent polarization is 'frozen' due to the permanent dipoles present in the dielectrics even without an external field. Such dielectrics have an asymmetry in their molecular structure and are described as 'polar'. |
On applying an external electric field, the dipoles arrange themselves according to the field lines. The effect of applied external field on the dipoles is determined by their dipole moments. Depending upon the bond between the positive and the negative charges in the molecules of a material, the orientation of dipoles takes place. The effective field intensity established in the material is the quantity induced by the external source and the sources within the material itself. The orientation of dipoles may be in the form of an arrangement in an element or they may simply straighten out like a chain, which usually depends upon the local field in which the molecules are situated. The local microscopic field may not be necessarily equal to the macroscopic applied electric field. |
| |