Water surface acts as if it is in tension! Due to this phenomenon, small objects "float" on the surface of a fluid, as long as the objects cannot break through and separate the top layer of water molecules. Small creatures such as “water strider” and “Basilisk Lizard” thus can walk on water as the exerted pressure is not sufficient to penetrate through the water surface as shown in Fig. 3.1a (http://norfolkwildgardens.co.uk/Pond-Skater.html) and 3.1b (http://www.factzoo.com/reptiles/lizards/jesus-lizard-running-on-water.html).
Surface Tension
Surface tension is due to an imbalance between intermolecular attractions (cohesive forces) at the surface. Water molecules hold each other due to the intermolecular cohesive forces. These forces acting on a water molecule, are effectively equal in all the directions in the bulk solution. Thus, the net force on the molecules in the bulk solution is zero. However, a resultant inward force acts on the molecules at the air-water interface (surface) due to the absence of the water molecule above, as shown in Fig. 3.2. The presence of fewer water molecules at the surface also results in a stronger bond between the molecules. This inward net force causes molecules at the surface to contract and to resist being stretched or broken. Thermodynamically, the molecules at the surface must gain excess energy over those in the bulk water to retain at the surface. This surface free energy is the surface tension at the air-water interface and the surface behaves like a tension membrane.
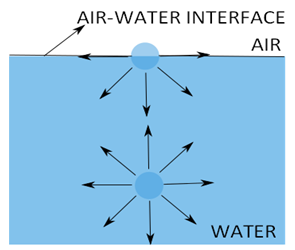
Fig. 2. Development of surface tension at the air-water interface