Sodium present in the adsorbed layer is normalized with respect to total exchange capacity as represented by Eq. 2.20 and is termed as exchangeable sodium percentage (ESP).
| ESP = ((Na+)s / (total exchange capacity)) X 100 |
2.20 |
ESP and SAR are considered to be a reliable indicator of clay stability against breakdown and particle dispersion especially for non-marine clays (Mitchell and Soga 2005). Clays with ESP > 2 percent is considered as dispersive.
Other quantitative attributes of cation exchange in soils is the property known as “the percentage base saturation” (Eq. 2.21), which denotes the measure of the proportion of exchangeable base on the soil exchange complex.
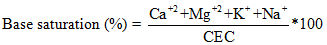 |
2.21 |
Factors influencing CEC of the soil
a. pH of the soil
It is observed that CEC of the soil increases with an increase in pH. Therefore, it is recommended to maintain a neutral pH (= 7.0) for determining CEC of the soil.
b. Presence of organic matter
The presence of organic carbon in clays reduces its CEC (Syers et al., 1970). However, some studies report an increase in CEC with increasing organic matter contents and this effect was more pronounced in coarser fractions.
c. Temperature
The ion exchange capacity decreases with an increase in temperature.
d. Particle size
It is observed that CEC increase with decreasing particle.
e. Calcium carbonate contents (CaCO3)
Higher amount of CaCO3 in soil leads to higher CEC.
f. Mineralogy
Active clay minerals increase CEC of the soil.
Determination of CEC by Ammonium replacement method
Horneck et al. (1989) have proposed a method for determining the CEC of soils by using ammonium replacement technique. This method involves saturation of the cation exchange sites on the soil surface with ammonium, equilibration, and removal of excess ammonium with ethanol, replacement and leaching of exchangeable ammonium with protons from HCl acid. It must be noted that this method is less suited for soils containing carbonates, vermiculite, gypsum and
zeolite minerals. The procedure is discussed as follows:
- Take about 10 g of soil, in 125 ml flask, add 50 ml of ammonium acetate solution and place the flask in reciprocating shaker for 30 min. The shaking process is repeated with blank solution as well.
- 1 liter vacuum extraction flask is connected to a funnel with Whatman no.5 filter paper. The soil sample is then transferred to the funnel and leached with 175 ml of 1 N ammonium acetate. The leached solution is analyzed for extractable K, Ca, Mg, and Na.
- The soil sample in the funnel is further leached with ethanol and the leachate is discarded.
- Transfer the soil to a 500 ml suction flask and leach the soil sample with 225 ml of 0.1N HCl to replace the exchangeable ammonium. Make up the leachate to a final volume of 250 ml in a standard flask using deionized water.
- The concentration of ammonium in the final leachate is measured, and CEC is calculated using Eq.
2.22.
 |
2.22 |
IS code (IS 2720, Part 24 1976) and USEPA (EPA SW-846) also provide
alternate methods for determining CEC of the soil. The range of CEC values for different soil minerals are listed in Table 2.3 (Carroll 1959). It can be noted that highly active soil minerals such as montmorillonite and vermiculite exhibit high CEC. Therefore, CEC is important in
assessing the chemical properties of the soil in terms of its reactivity, contaminant retention mechanism etc.
| Table 2.3 CEC values of common soil minerals |
| Mineral |
CEC (meq./100g) at pH 7 |
| Kaolinite |
3-15 |
| Illite |
10-40 |
| Montmorillonite |
70-100 |
| Vermiculite |
100-150 |
| Halloysite 2H2O |
5-10 |
| Halloysite 4H2O |
40-50 |
| Chlorite |
10-40 |
| Allophane |
60-70 |