Cation exchange capacity
From previous discussion, it is clear that clay surface adsorbs specific amount and type of cations under a given environmental conditions such as temperature, pressure, pH and pore water chemistry. The adsorbed cations can get partly or fully replaced by ions of another type subject to changes in the environmental condition. Such changes can alter the physico-chemical characteristics of soil. The most common cations present in the soil are sodium, potassium, calcium and magnesium. Marine clays and saline soils contain sodium as the dominant adsorbed cation. Acidic soils contain Al+3 and H+. The most common anions are sulphate, chloride, phosphate and nitrate.
Cation exchange capacity (CEC) is defined as the sum of exchangeable cations soil can adsorb per 100 g of dry soil. Its unit is meq./100 g and normally its value ranges between 1 and 150 meq./100 g. The value represents the amount of exchangeable cations that can be replaced easily by another incoming cation. The replaceability of cations depends on valency, relative abundance of different ion type and ion size. All other factors remaining same, trivalent cations are held more tightly than divalent and univalent ions. A small ion tends to replace large ions. It is also possible to replace a high replacing power cation by one of low replacing power due to the high concentration of latter in the pore solution. For example: Al+3 can be replaced by Na+
due to its abundance. A typical replaceability series is given as follows: (Mitchell and Soga 2005)
Na+ < Li+ < K+ < Cs+ < Mg+2 < Ca+2 < Cu+2 < Al+3 < Fe+3
In kaolinite, the exchange reaction takes place quickly. In illite, a small part of the exchange sites may be between unit layers of minerals and hence would take more time. In smectite minerals, much longer time is required because the major part of exchange capacity is located in the interlayer region.
For a pore solution containing both monovalent and divalent cations, the ratio of divalent to monovalent cations is much higher in adsorbed layer than in the equilibrium solution. If M and N represent monovalent cation concentrations, P the concentration of divalent ions, subscript s and e represent adsorbed ions on soil and that in equilibrium solution, respectively, then
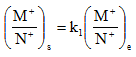 |
2.17 |
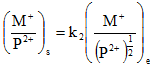 |
2.18 |
where k1 and k2 are selectivity constants, which can be obtained experimentally. Following, Eq. 2.18 it can be further written as
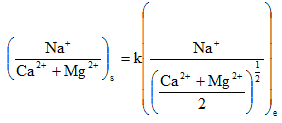 |
2.19 |
The concentration of cation is in milliequivalents per litre. The quantity 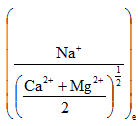 is termed as sodium adsorption ratio (SAR) in (meq./litre)1/2. If the composition of pore fluid and k is known, the relative amounts of single and divalent cations in the adsorbed cation complex can be determined. The details of selectivity constants for a wide variety of clays are reported by Bruggenwert and Kamphorst (1979).
is termed as sodium adsorption ratio (SAR) in (meq./litre)1/2. If the composition of pore fluid and k is known, the relative amounts of single and divalent cations in the adsorbed cation complex can be determined. The details of selectivity constants for a wide variety of clays are reported by Bruggenwert and Kamphorst (1979).