Iron Proteins:
Iron containing proteins are mainly belonging to four categories.
- Iron-porphyrin proteins: hemoglobin, myoglobin, cytochrome P450, etc; all of them contains one of more iron-porphyrin units. They are mainly involved in oxygen transfer, oxygen storage, and electron transfer.
- Non-heme iron proteins: ferritin, transferrin, hemesiderin, etc; they are mostly involved in iron storage and transport
- Non-heme diiron oxo-bridged species: ribonucleotide reductase, hemerythrin, methane monooxygenase, etc.
- Non-heme iron-sulfur cluster proteins: nitrogenase, ferredoxins, rubredoxins. They are mainly involved in biological electron transfer reactions.
Herein, some of the important iron proteins will be discussed.
Hemoglobin and Myoglobin:
Hemoglobin is an essential iron protein for molecular oxygen (dioxygen) transport and found in red blood cells. Both are globular proteins. Myoglobin is engaged in storage of molecular oxygen in muscle tissues and controlled transport of molecular oxygen for the oxidative reactions.
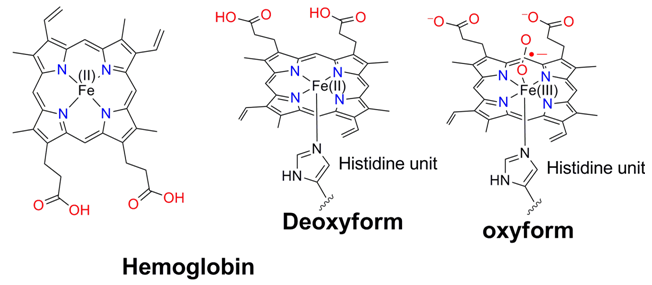
A hemoglobin unit is composed of has four protein chains each of which contains one porphyrin ring coordinated to iron (known as heme) packed in a roughly tetrahedral α2β2 cluster. The a unit contains 141 amino acids residue and the b unit contains 146 amino acids residue.
In hemoglobin, a high-spin Fe(II) is coordinated to four N atoms of porphyrin ring. The fifth coordination site is occupied by a histidine group. In this condition the protein containing four heme compartments is in stain condition and called tense (T) state. After binding to molecular oxygen high-spin Fe(III) changes to low-spin Fe(III) and molecular oxygen is transferred to superoxide. Covalent radii of Fe(II) is too large to fit into the cavity created by four N atoms of the porphyrin ring. Formation of low-spin Fe(III), the radii of iron decreases. Moreover, transformation of square pyramidal deoxy form to octahedral oxy form leads ion center closer to heme cavity. The protein is now in comparatively less stain relaxed form (R). In this was all the four iron centers in hemoglobin are transferred to oxo-hemoglobin forms.
Binding of molecular oxygen to the iron center of the hemoglobin tetramer causes release of protons from the acid units which minimize the pH. This lowering of pH favors molecular oxygen release to tissues and conversion of Fe(III) to Fe(II). This deoxyhemoglobin picks up 2 protons and 2 molecules of CO2 form tissues and carried to the lungs, where the CO2 is released. After that, deoxyhemoglobin which further binds to molecular oxygen and the O2 carrying and CO2 returning processes from tissues continue.
Myoglobin is a single chain heme protein containing 154 amino acids and it contains several region of α–helix. The structure of the active site (where the reaction occurs) and the oxygen carrying mechanism is same as hemoglobin. The oxygen uptake capacity of myoglobin is thermodynamically more compared to hemoglobin and hence, oxygen uptake in myglobin is more than that of hemoglobin.