Artificial radioactivity:
Bombardment of stable nuclei with high energy a -particle, b -particle, neutrons, deuterons or g -rays generates radioactive nuclides. Generation of this kind of radioactive material from sable material is called artificial radioactivity creation.
Examples:
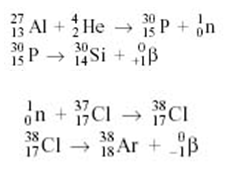
Carbon dating:
Carbon (C) atom has three naturally occurring isotopes. Both 12C and 13C are stable while, 14C is radioactive and decays by very weak beta decay to 14N with a half-life of approximately 5,730 years. Naturally occurring radioactive carbon is produced as a secondary effect of cosmic-ray bombardment of the upper atmosphere. The radioactive carbon is oxidized to carbon dioxide. Plants up take this carbon dioxide from the atmospheric and consequently, radioactive carbon enters into the food chain. Animals eat the plants and introduce carbon into their bodies. The intake of this radioactive carbon into the body cease as soon as body of plants or animal dies.
![]()
The radioactive carbon in the tissue decays continuously. The measurement of the remaining activity in the body provides a method of age determination of plants or animals.
The application of the 14C dating is based on the assumption that the intensity of cosmic ray as well as 14C remaining constant over thousands of years. This assumption implies that the radioactivity in a living object is same as the activity of any living object in many thousands years ago.
Hence,
Age = [(2.303×5730)/0.693]log(activity of the recent object/activity of the examined sample) a
The half life time ( t ½ ) of 4C = 5730 a (a stands for years)
Q. A specimen sample containing 14C gives 60 counts in 10 minutes per gram of carbon. A specimen of a freshly cut wood gives 10 counts per gram of carbon in a minute. The back ground count is 5 per minutes in absence of any 14C containing sample. What is the age of the specimen sample?
Ans:
The count rate of the specimen = {(60/10) - 5} = 6 - 5 = 1
The count rate of the fresh wood = 10 - 5 = 5
Therefore,
Age = [(2.303×5730)/0.693] log (5/1) a = 13310 a